
Pooja Choradia: vWF Breakdown in Aortic Stenosis – Anaemia and Pulmonary Relevance
Pooja Choradia, Critical Care Associate at P.D. Hinduja Hospital Khar, shared a post on LinkedIn:
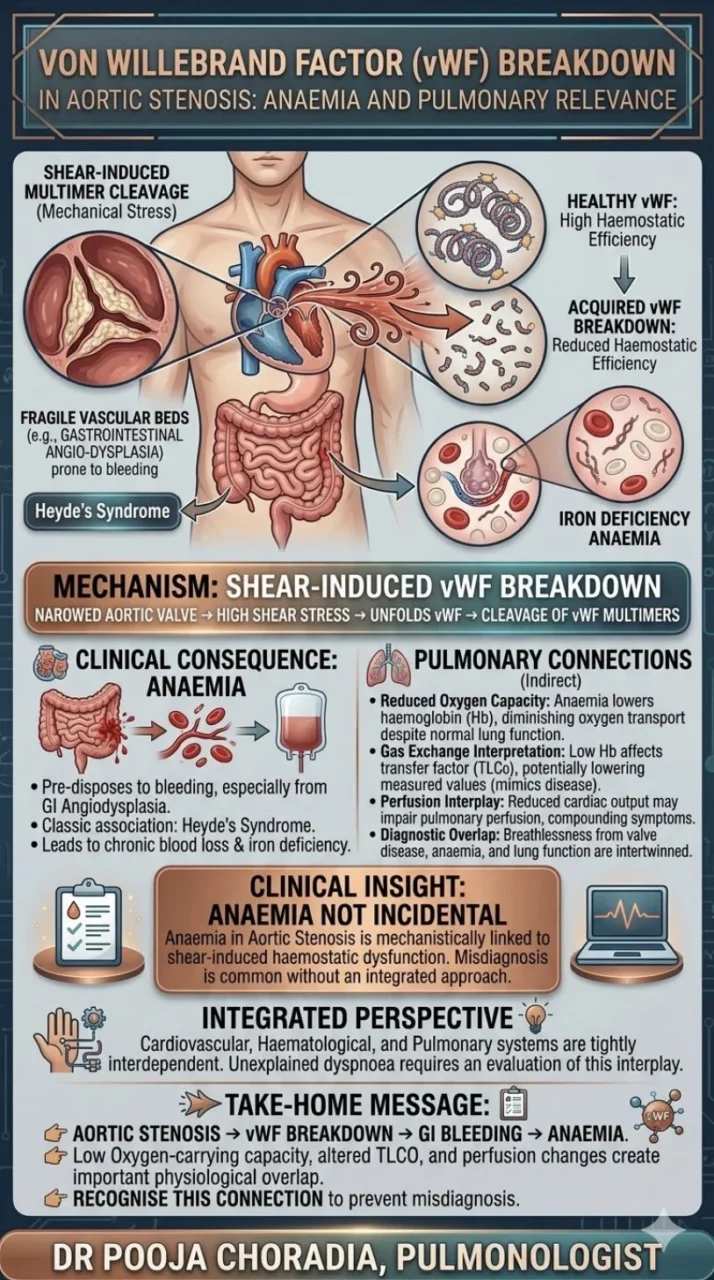
“Von Willebrand Factor Breakdown in Aortic Stenosis: Anaemia and Pulmonary Relevance
Severe aortic stenosis is not solely a valvular outflow disorder—it also has important haematological consequences. One of the most significant is the acquired disruption of von Willebrand factor (vWF), leading to bleeding tendency and, frequently, anaemia.
Mechanism: Shear-Induced vWF Breakdown
In aortic stenosis, blood is forced through a narrowed valve at high velocity, generating excessive shear stress. This mechanical stress unfolds large vWF multimers, making them highly susceptible to cleavage. As a result, the most functionally important high-molecular-weight vWF multimers are lost.
This produces an acquired form of von Willebrand syndrome, characterised by impaired platelet adhesion and reduced haemostatic efficiency.
Clinical Consequence: Anaemia
The breakdown of vWF predisposes to bleeding, particularly from fragile vascular beds such as gastrointestinal angiodysplasia. This association—classically described in Heyde’s syndrome—leads to chronic blood loss and iron deficiency anaemia.
Thus, the anaemia seen in aortic stenosis is not simply incidental; it is mechanistically linked to shear-induced haemostatic dysfunction.
Is There a Pulmonary Connection?
The relationship to lung physiology is indirect but relevant:
Reduced Oxygen Carrying Capacity: Anaemia lowers haemoglobin levels, diminishing oxygen transport despite normal lung function. This can exacerbate dyspnoea, often attributed solely to cardiac or pulmonary causes.
Gas Exchange Interpretation: In physiological testing, reduced haemoglobin affects transfer factor (TLCO), potentially lowering measured values. This may mimic or obscure underlying pulmonary vascular or interstitial disease if not corrected for haemoglobin.
Perfusion Interplay: Severe aortic stenosis alters systemic and pulmonary haemodynamics. Reduced cardiac output may impair pulmonary perfusion, compounding symptoms without primary lung pathology.
Diagnostic Overlap: Patients may present with breathlessness, where contributions from valvular disease, anaemia, and pulmonary function are intertwined. Without integrating these factors, misinterpretation is common.
Integrated Perspective
This condition highlights a broader principle: cardiovascular, haematological, and pulmonary systems are tightly interdependent. A primary valvular lesion can lead to haemostatic dysfunction, which in turn alters oxygen delivery and influences respiratory assessment.
Understanding this interplay is essential, particularly when evaluating unexplained dyspnoea or abnormal gas transfer results.
Conclusion
In aortic stenosis, shear-induced breakdown of von Willebrand factor leads to bleeding and anaemia, most notably in the context of gastrointestinal angiodysplasia.
While the lung is not directly involved, the downstream effects—reduced oxygen carriage, altered TLCO, and perfusion changes—create important physiological overlap.”
Stay updated with Haemostasis Today.
-
May 23, 2026, 03:02Maitri Vaishnav: Anatomical Variability and Surgical Relevance of Corona Mortis
-
May 23, 2026, 02:26Muhammad Hashim: When the Blood Smear Told a Different Story Behind a ‘Critical’ Platelet Count
-
May 23, 2026, 02:20Augustina Isioma Ikusemoro: Understanding One of the Most Common and Preventable Transfusion Reactions
-
May 23, 2026, 02:15What’s Your Advice for the Newly Diagnosed? – ITP Support Association
-
May 23, 2026, 02:08Rob Maloney: When You Stop Building Only a Career and Start Building a Life of Significance
-
May 23, 2026, 02:04Rahmathullah S.N.: Key Takeaways on Successful Surgical Outcomes in Patients with Congenital Bleeding Disorders
-
May 23, 2026, 01:51Paula Greter: Access and Innovation in Hemophilia through ERAH2026
-
May 22, 2026, 17:30Veronica Sanchez: Do You Live with Pain after a Stroke?
-
May 22, 2026, 17:25Maxime Dely: A Simple but Powerful Indicator of Platelet Quality

