Stan Van Boeckel: Pitfalls in Early Drug Discovery
Stan Van Boeckel, Professor Emeritus at Leiden University, shared a post on LinkedIn:
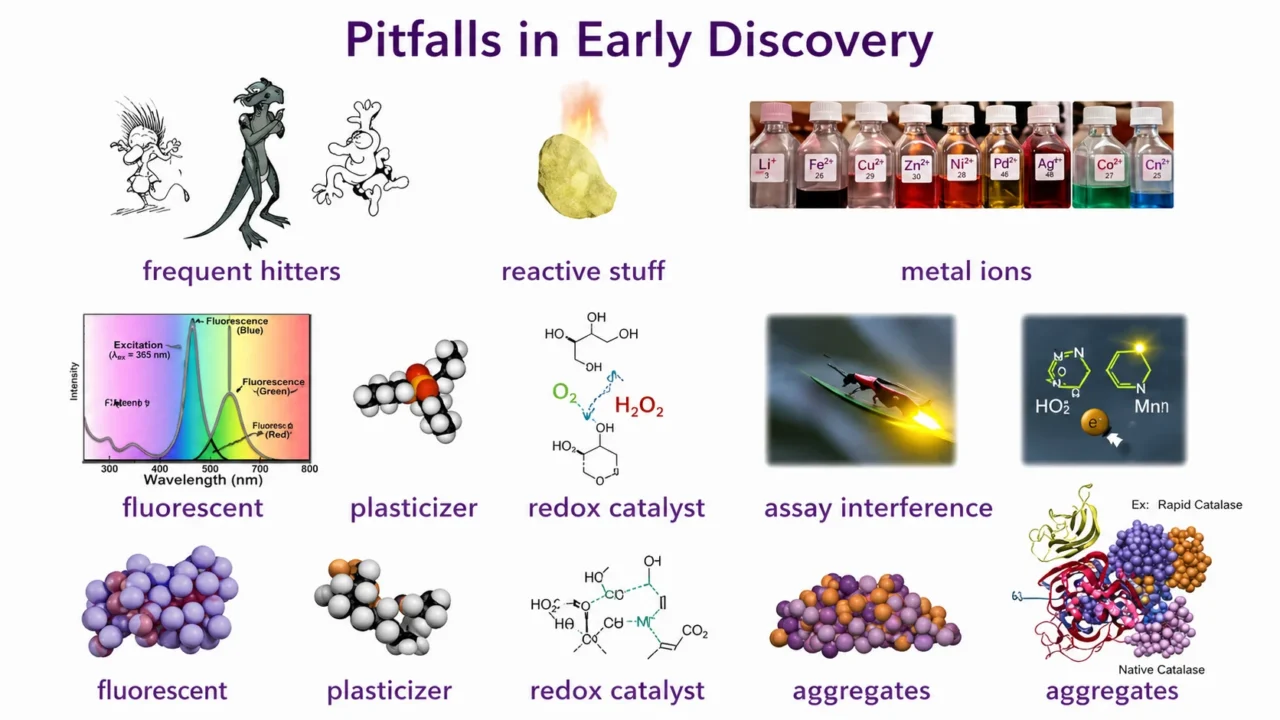
”Part 30 — Pitfalls in Early Drug Discovery.
After more than forty years in medicinal chemistry across Pharma, Biotech, and Academia, I have repeatedly encountered misleading results, especially in early research where sensitive in‑vitro assays dominate.
Unfortunately, the same problems keep resurfacing. I still recall, four decades ago, working on inositol‑phosphate analogues to inhibit platelet aggregation. One compound looked spectacular, but after resynthesis and further purification the activity vanished.
The original batch contained a plasticizer-tributyl phosphate-which turned out to be the true active.
Since then, impurities have remained firmly on my radar: residual metal ions, reactive species, coloured or fluorescent compounds, intermediates, by‑products, stabilizers, plasticizers. They reappear in every generation of research, and investigators often begin in a phase of denial.
I have repeatedly met enthusiastic scientists convinced they had discovered unexpectedly potent hits or leads, only to learn that the compound adhered to the pipette tip and produced artificially high concentrations upon serial dilution. Sometimes the compound itself is the problem.
Entire classes of molecules are notorious for showing activity in many assays, and several are now well documented as PAINS. Others interfere with specific assay formats: luciferase inhibitors, fluorescent molecules, compounds that catalyse redox cycling and generate hydrogen peroxide in the presence of DTT.
Some react nonspecifically with the target through metal coordination or covalent modification. And perhaps the most misleading of all are aggregating compounds.
I have seen many papers where I wondered whether the reported inhibition was simply due to aggregates.
Large lipophilic or amphiphilic molecules are suspect, and repeating dose-response curves in the presence of non‑ionic detergents or higher protein concentrations should settle the question, yet this control is often omitted.
A striking example was published by Janssen R and D: a supposed lead that disrupted the protein–protein interaction between TNF‑α and its receptors.
Selective inhibitors of protein–protein interactions are difficult to find anyway so the team was cautious. Their ‘lead’ turned out to form a large, remarkably stable, ordered pentameric aggregate that interfered with binding, and remarkably this aggregate was still visible in the crystal structure with the protein.”
Stay updated with Hemostasis Today.
-
Apr 28, 2026, 15:47Dianne E. van der Wal: How Rat and Human Platelet Vesicles Are Different
-
Apr 28, 2026, 15:14Elaheh Mansouri: Platelet Clumping Is a Common Pre-Analytical and Analytical Challenge in Haematology
-
Apr 28, 2026, 15:11Hans Vrielink: Honoured to Receive the Cohn the Laval Award at the ASFA/WAA
-
Apr 28, 2026, 15:02Stephani Carolina Salvatierra Moreno: Our Study on Microneedling and PRP Has Been Published
-
Apr 28, 2026, 14:57Steven Heylen: Hydrolyzed Collagen Injections for Knee Osteoarthritis as a New Treatment Option
-
Apr 28, 2026, 14:22Heghine Khachatryan: Regarding Coagulation Factor Dynamics During Pregnancy in Hemophilia B Carriers
-
Apr 28, 2026, 14:20Ney Carter Borges: Controlling Thrombosis Without Increasing Bleeding Risk – A New Frontier in Antiplatelet Therapy
-
Apr 28, 2026, 14:20Manoj Kumar Singh: Participation in Research Is the Responsibility for a Healthier India
-
Apr 28, 2026, 14:19UK NEQAS for Blood Coagulation: Providing an Opportunity to Showcase Your Work at CLH 2026

