
Transforming FVIII Product Classification: AUC and TTR as Game-Changing Metrics for Next-Generation Hemophilia A Therapy
Wolfgang Miesbach, Professor of Medicine at Frankfurt University Hospital, has recently shared a very informative post on X:
‘‘Novel FVIII Classification: Scientific Framework for Next-Generation Haemophilia Treatment. Two pharmacokinetic metrics that can redefine how we classify Factor VIII products, presented by Professor Cedric Hermans at ISTH SSC FVIII/FIX session.
The Scientific Challenge
Traditional “standard” vs “extended” half-life classifications have proven inadequate for new generation FVIII products. Recent advances have led to improved ability to maintain FVIII levels within normal range.
New Classification Metrics
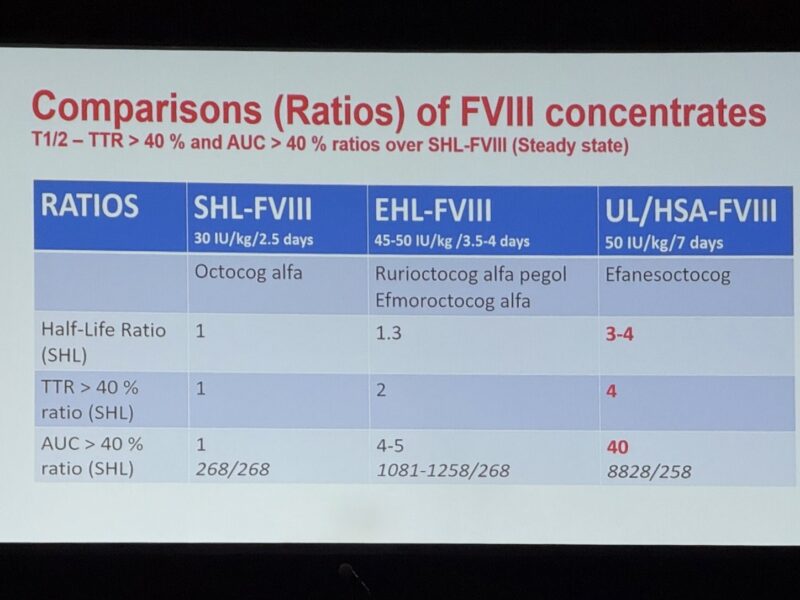
– Area Under the Curve (AUC) > 40%
Measures total drug exposure over time, providing comprehensive assessment of sustained FVIII activity rather than peak-focused measurements
– Time in Target Range (TTR) > 40%
Evaluates percentage of time patients maintain therapeutically effective FVIII levels, offering crucial insights into sustained haemostatic protection
Comparative Performance Data
– Standard Half-Life (SHL-FVIII): Baseline comparators (AUC ratio: 1, TTR ratio: 1)
– Extended Half-Life (EHL-FVIII): 4-5x AUC improvement, 2x TTR enhancement
– Ultra-Long/High Sustained Activity (UL/HSA-FVIII): 40x AUC improvement, 4x TTR boost
Products like efanesoctocog alfa demonstrate remarkable sustained activity, maintaining mean FVIII levels >40 IU/dL for 4 days with geometric mean half-life of 43.3 hours
Future Integration
Further validation can be warranted for integration into routine clinical evaluation and PK tools like WAPPS-Hemo, MyPKFit, and Florio. These platforms enable Bayesian forecasting of individual PK profiles for personalized treatment optimization.
As the field evolves toward normal hemostasis as the treatment goal, these metrics provide an important scientific basis for evaluating innovative therapies.”
Stay updated with Hemostasis Today.
-
May 27, 2026, 17:51Nita Radhakrishnan: Post Graduate Institute of Child Health Receives WFH Data Quality Certification for Excellence
-
May 27, 2026, 17:51Fiona Robinson: What exactly Are Rebalancing Agents for Bleeding Aisorders?
-
May 27, 2026, 17:14Khurram Nasir: Can Earlier LDL-C Lowering Change the Trajectory of Cardiovascular Disease?
-
May 27, 2026, 17:09Yvonne Jongejan: Allele-Selective Silencing Therapy for VWD and Thrombotic Disorders
-
May 27, 2026, 16:58Catherine Moran: Important Step Forward as 1st WHO Resolution on Stroke Gets Adopted at WHA79
-
May 27, 2026, 16:08Heghine Khachatryan: Thrombosis in Oncology Remains a Major Healthcare Burden
-
May 27, 2026, 16:00Revealing Critical Gaps in Hemophilia Diagnosis and Access to Care in India – WFH
-
May 27, 2026, 16:00Melissa Hollo: Women Deserve Proactive Care, Not Reactive Care
-
May 27, 2026, 15:59Piotr Czempik: First Report of ROTEM Findings in Triple-Positive APS and Hageman Anomaly

