
Lucia Masarova/X
Sep 5, 2025, 12:18
Lucia Masarova Presents Results of Phase III SURPASS-ET Trial at SOHO 2025: Ropeg’s Efficacy for Essential Thrombocythemia
MPN Hub shared on X:
”CONGRESS | SOHO 2025 | POSTER
Lucia Masarova, MD Anderson Cancer Center
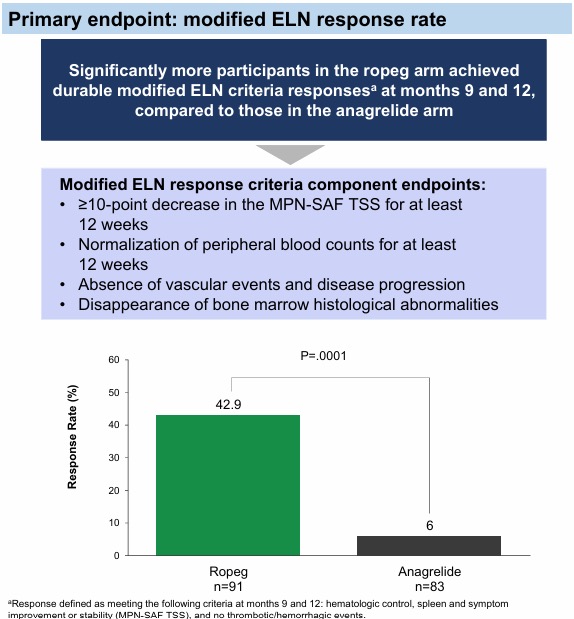
Presents results from the phase III SURPASS-ET trial of ropeginterferon alfa-2b (n=91) vs anagrelide (n=83) in patients with HR Essential Thrombocythemia who are resistant or intolerant to hydroxyurea.
Ropeg showed superior efficacy over anagrelide (RR, 42.9% vs 6.0%, P=0.0001) and demonstrated a favorable safety profile.”
All on Hemostasis and Thrombosis from SOHO 2025 featured in Hemostasis Today.
-
Apr 21, 2026, 21:07Tareq Abadl: Not Every Low Platelet Count Is True – Check the Smear First
-
Apr 21, 2026, 21:05Beverley Hunt: Honored to Receive the Lifetime Achievement Award at BSH 2026
-
Apr 21, 2026, 21:03Toong Youttananukorn: Proud to Support Data That Drives Action at WFH 2026
-
Apr 21, 2026, 20:48Ney Carter Borges: Endothelial Senescence as a Central Driver of Adipose Dysfunction in Heart Failure and Type 2 Diabetes
-
Apr 21, 2026, 20:29Can Stem Cell-Delivered Platelets Fight Drug-Resistant Bacteria?- RPTH Journal
-
Apr 21, 2026, 18:51WHO Expert Lorenzo Moja Discusses How Essential Medicines Policy Expands Access to Treatment – WFH
-
Apr 21, 2026, 18:35Ana Pedrero Gil: New ASH–ISTH 2026 Guidelines Focus on Preventing VTE in Children
-
Apr 21, 2026, 17:36Sip Wijchers: Is It Time to Use Ultrasound for Every AF Ablation Access?
-
Apr 21, 2026, 16:32Sarah Walbolt։ Have You Ever Wondered What Actually Happens to Your Donated Blood?

