
Ney Carter Borges: Endothelial Senescence as a Central Driver of Adipose Dysfunction in Heart Failure and Type 2 Diabetes
Ney Carter Borges, Member Cardiologist of Global Physician Association at Cleveland Clinic Florida, shared a post on LinkedIn:
“Endothelial Senescence as a Central Driver of Adipose Dysfunction in Heart Failure and Type 2 Diabetes:
Therapeutic Implications of Senolytic Targeting
This translational study investigates the role of endothelial senescence in subcutaneous adipose tissue (SAT) dysfunction among patients with heart failure and type 2 diabetes (HFDM).
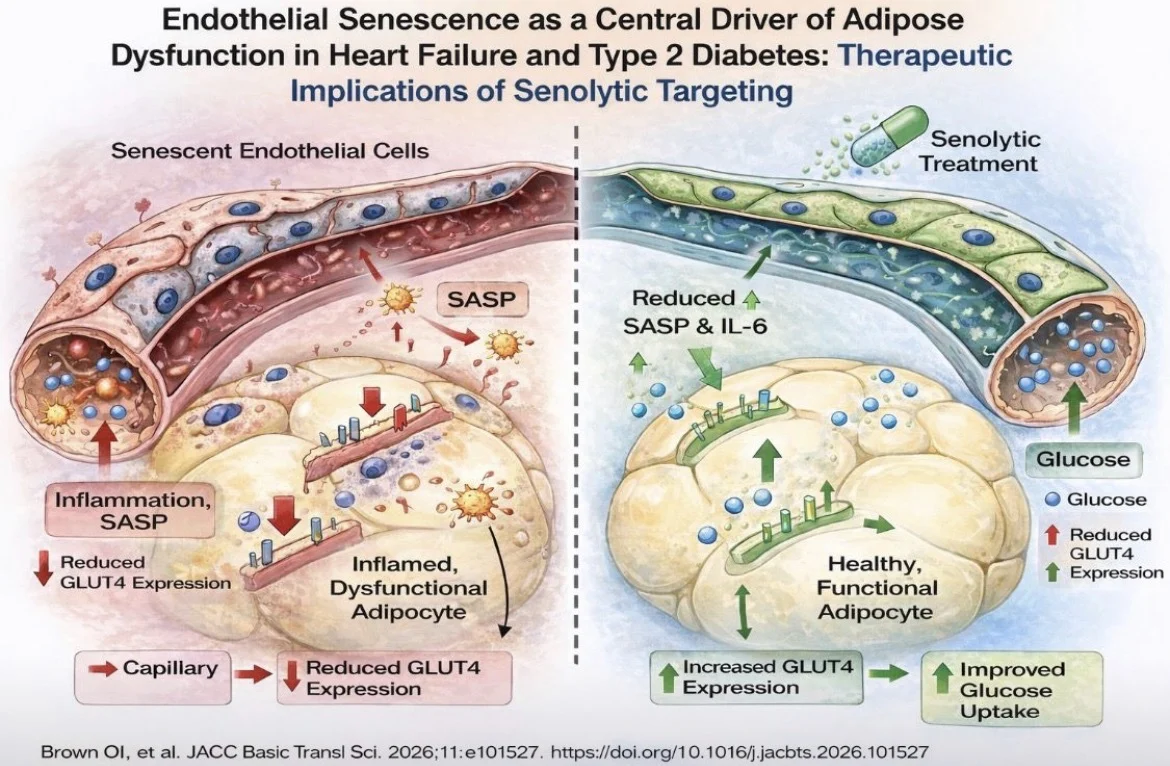
Using human SAT biopsies, the authors demonstrate that microvascular endothelial cells (MVECs) from HFDM patients exhibit a clear senescent phenotype, characterized by increased senescence-associated β-galactosidase activity, reduced proliferative and angiogenic capacity, mitochondrial dysfunction, and a proinflammatory secretory profile (SASP).
Functionally, these senescent endothelial cells drive maladaptive cross-talk with adipocytes.
In coculture experiments, they induce a proinflammatory adipocyte phenotype marked by increased interleukin-6 expression, reduced adiponectin, impaired GLUT4 expression, and decreased glucose uptake—key features of metabolic dysfunction.
This establishes endothelial senescence as a central mechanistic link between vascular dysfunction and impaired adipose metabolic regulation in HFDM.
Importantly, the study identifies digoxin, at therapeutic concentrations, as a potential senolytic agent.
Treatment with digoxin significantly reduced endothelial senescence markers, improved mitochondrial function and proliferation, and attenuated inflammatory cytokine secretion.
Consequently, adipocytes exposed to digoxin-treated endothelial cells demonstrated improved glucose uptake and a more favorable metabolic phenotype.
These findings introduce a novel paradigm in cardiometabolic disease:
Endothelial senescence is not merely a bystander but an active driver of adipose tissue dysfunction.
Therapeutically targeting senescent endothelial cells may therefore represent a promising strategy to restore metabolic homeostasis in high-risk HFDM populations.
However, clinical translation requires caution given digoxin’s narrow therapeutic window and the need for in vivo validation.
Overall, this study provides strong mechanistic insight and opens new avenues for senescence-targeted therapies in cardiometabolic disease.”
More posts featuring Ney Carter Borges on Hemostasis Today.
-
Apr 21, 2026, 20:29Can Stem Cell-Delivered Platelets Fight Drug-Resistant Bacteria?- RPTH Journal
-
Apr 21, 2026, 18:51WHO Expert Lorenzo Moja Discusses How Essential Medicines Policy Expands Access to Treatment – WFH
-
Apr 21, 2026, 18:35Ana Pedrero Gil: New ASH–ISTH 2026 Guidelines Focus on Preventing VTE in Children
-
Apr 21, 2026, 17:36Sip Wijchers: Is It Time to Use Ultrasound for Every AF Ablation Access?
-
Apr 21, 2026, 16:32Sarah Walbolt։ Have You Ever Wondered What Actually Happens to Your Donated Blood?
-
Apr 21, 2026, 16:08Piotr Czempik: Hidden Platelet Dysfunction in Sepsis and Septic Shock
-
Apr 21, 2026, 16:04Akshat Jain: Personalized Hemophilia Care in the Era of Novel Therapies
-
Apr 21, 2026, 16:03Abdelaziz Fahem։ Novo Nordisk Reports Positive Phase 3 Results for Etavopivat
-
Apr 21, 2026, 16:01Augustina Isioma Ikusemoro: ABO Discrepancies in Transfusion Practice

