
Vikas Londhe on Andexxa’s Safety Assessment Updates
Vikas Londhe, Chief Editor of Pharmacally.com, shared on LinkedIn:
”FDA Safety Update: Andexxa Risks Outweigh Benefits After Postmarketing Thromboembolic Events—Market Withdrawal Planned
FDA Updates Safety Assessment of Andexxa After Serious Thromboembolic Events.
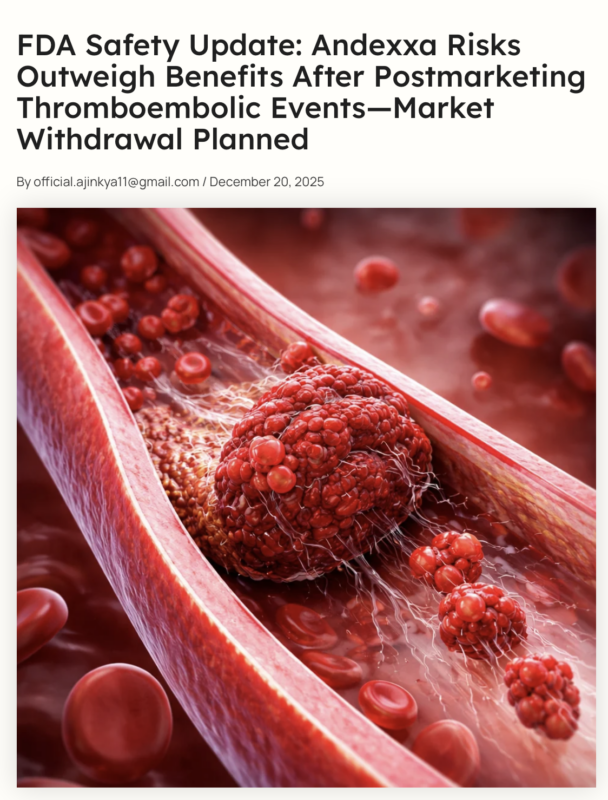
On December 18, 2025, the U.S. Food and Drug Administration (FDA) issued a safety communication regarding Andexxa (coagulation factor Xa (recombinant), inactivated-zhzo).
The agency has reviewed postmarketing safety data showing an increased incidence of thromboembolic events, including serious and fatal outcomes, in patients treated with Andexxa.
Based on these findings, the FDA now considers that the risks associated with Andexxa outweigh its benefits in its approved use.”
Read the full announcement here.
Stay updated with Hemostasis Today.
-
May 24, 2026, 17:13Mahtab Mashayekhi: The Evolving Role of Red Blood Cell Exchange in the Management of SCD
-
May 24, 2026, 17:12Silvina Graña: Helping Families Better Understand Hemophilia and Daily Life With Bleeding Disorders
-
May 24, 2026, 17:11Miikka Vikkula: Key Insights from the ISSVA 2026 World Congress
-
May 24, 2026, 17:10Mathieu Jackson: Proud to Be Present at This Historic Moment for the Global Bleeding Disorders Community
-
May 24, 2026, 17:09Ramina Fazeli: Highlighting the Emerging Role of Neutrophil Extracellular Traps in Ovarian Cancer
-
May 24, 2026, 16:12William Wallace: Normal B12 Levels May Still Cause Brain Damage and Cognitive Decline
-
May 24, 2026, 15:54Marc Carrier: Ottawa Race Weekend Raised Over 7,000 USD for Thrombosis Canada
-
May 24, 2026, 15:44Janak Sadhu: Building a Stroke-Free Mehsana Through Community Awareness and Healthcare Collaboration
-
May 24, 2026, 15:33Mohammed Zubair: AI-Driven Innovation in Stroke Diagnosis and Medical Imaging

