
William Wallace/LinkedIn
Oct 23, 2025, 09:59
William Wallace: How Collagen Builds the Body’s Structural Framework
William Wallace, Director of Product Development and Scientific Affairs at ProHealth Longevity, shared a post on LinkedIn:
“How collagen builds the body’s structural framework
Collagen makes up nearly two-thirds of all the protein in the human body, acting as the biological scaffolding that gives skin, bone, cartilage, and connective tissue their strength and resilience. It’s not a single protein but a family of more than 30 types, each tailored to a specific tissue.
- From molecule to fiber
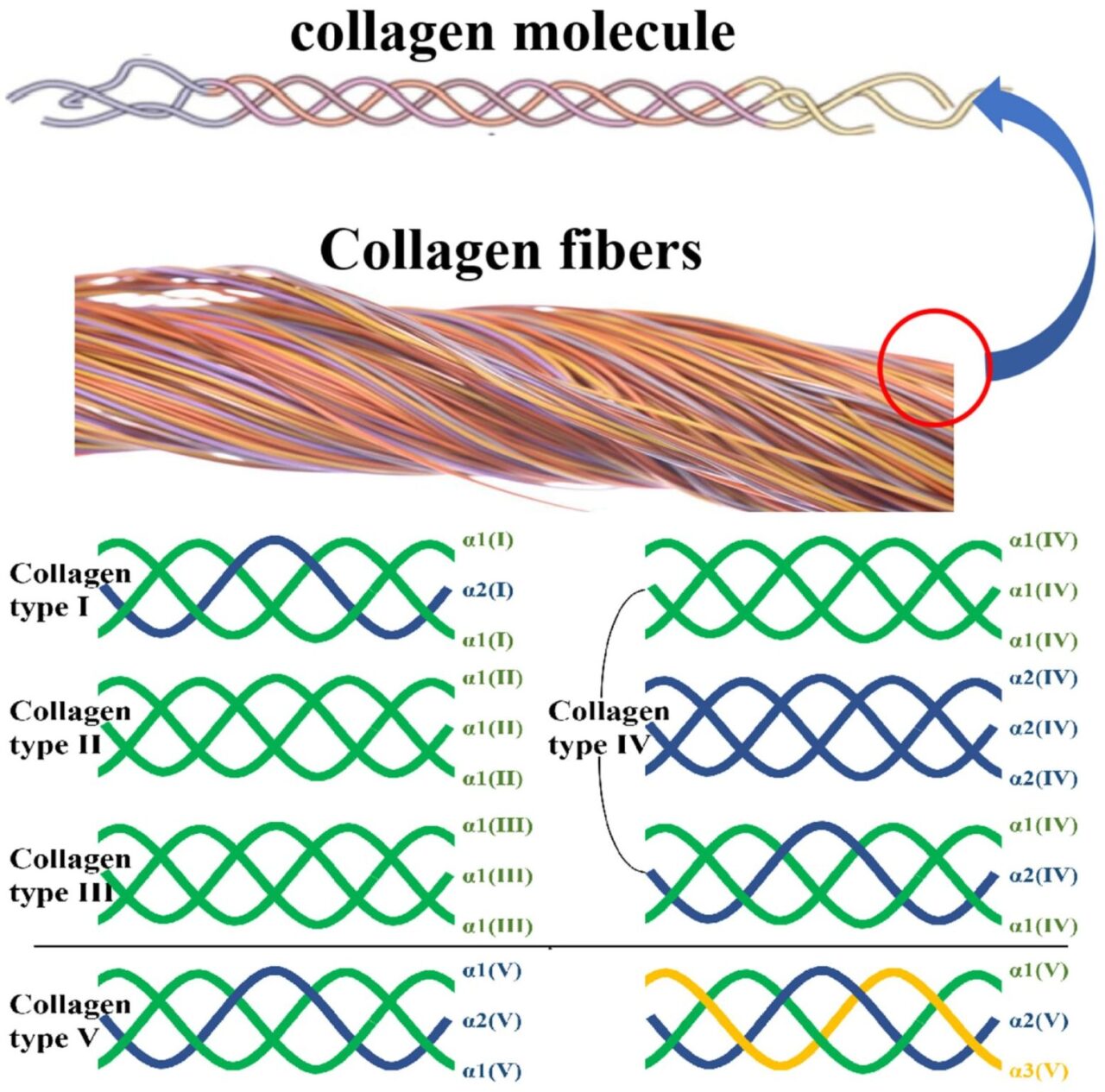
Every collagen molecule is a triple helix formed by three polypeptide chains (α-chains). Thousands of these helices bundle into fibrils, which then weave into collagen fibers—the cables that hold tissues together.
* Example: In tendons and ligaments, tightly packed type I collagen fibers resist stretching while still allowing flexibility under load. - Collagen type I — the dominant form
Type I collagen accounts for ≈90 % of the collagen in skin, ≈80 % of that in bone, and the majority in tendons and ligaments. It’s built from two α1(I) chains and one α2(I) chain, creating the strongest fibrillar form.
* Example: Its dense crosslinking provides tensile strength to the dermis and structural rigidity to bones. - Collagen type II — cartilage and joints
Type II collagen consists of three identical α1(II) chains and forms a fine mesh that cushions and stabilizes joints.
* Example: It’s the main structural protein of articular cartilage, giving flexibility while resisting compression. - Collagen type III and V — elasticity and regulation
Type III (three α1(III) chains) supports pliability in skin, blood vessels, and organs, often pairing with type I. Type V helps control fiber diameter and collagen organization, especially in the cornea, placenta, and skin.
* Example: Type III is abundant in arteries, allowing them to expand and recoil with blood flow. - Collagen type IV — the structural mesh
Type IV forms a non-fibrillar network in basement membranes, anchoring cells and filtering molecules in tissues like the kidney glomerulus and alveoli.
* Example: Its flexible lattice supports epithelial and endothelial layers without sacrificing permeability. - Nutrient support and stability
Vitamin C, copper, zinc, and iron are critical for hydroxylation and crosslinking reactions that stabilize collagen helices.
* Example: In vitamin C deficiency (scurvy), defective crosslinking leads to fragile skin, bleeding gums, and impaired wound healing.
From molecular helices to visible fibers, collagen’s hierarchy explains its dominance in the human body—it’s the primary structural protein that unites strength, flexibility, and repair across every organ system.”
Stay updated with Hemostasis Today.
-
May 24, 2026, 13:10Thomas Pincez: Emerging Challenges in Sickle Cell Disease Gene Therapy and Cancer Risk
-
May 24, 2026, 12:04Kalpana Gupta Shekhawat: Ferritin Is More Than an Iron Marker
-
May 24, 2026, 11:50Peter Zdziarski: Making Rare Disease Research More Human
-
May 24, 2026, 11:39Bram Berkelmans: Donating with a View
-
May 24, 2026, 11:28Ratiporn Chinthammit: A Meaningful Exchange in aHUS and PNH Education
-
May 24, 2026, 11:17Mary Ann Sromoski: Transfusion Medicine and Patient Blood Management in Practice
-
May 24, 2026, 07:45Thierry Burnouf: Advances in Platelet Membrane-Coated Nanoparticles for Targeted Therapeutics and Regenerative Medicine
-
May 24, 2026, 07:39Shamala Thilarajah: Our Team Receives the NMRC Transition Award for NEUROACT-Stroke
-
May 24, 2026, 05:48Yazan Abou-Ismail: Contributing to the Future of von Willebrand Disease Management

