Chokri Ben Lamine: IV Iron Showdown – FCM vs Ferric Derisomaltose vs Iron Dextran
Chokri Ben Lamine, Assistant consultant at King Faisal Specialist Hospital and Research Center, shared a post on X:
“IV iron showdown – FCM vs ferric derisomaltose vs iron dextran
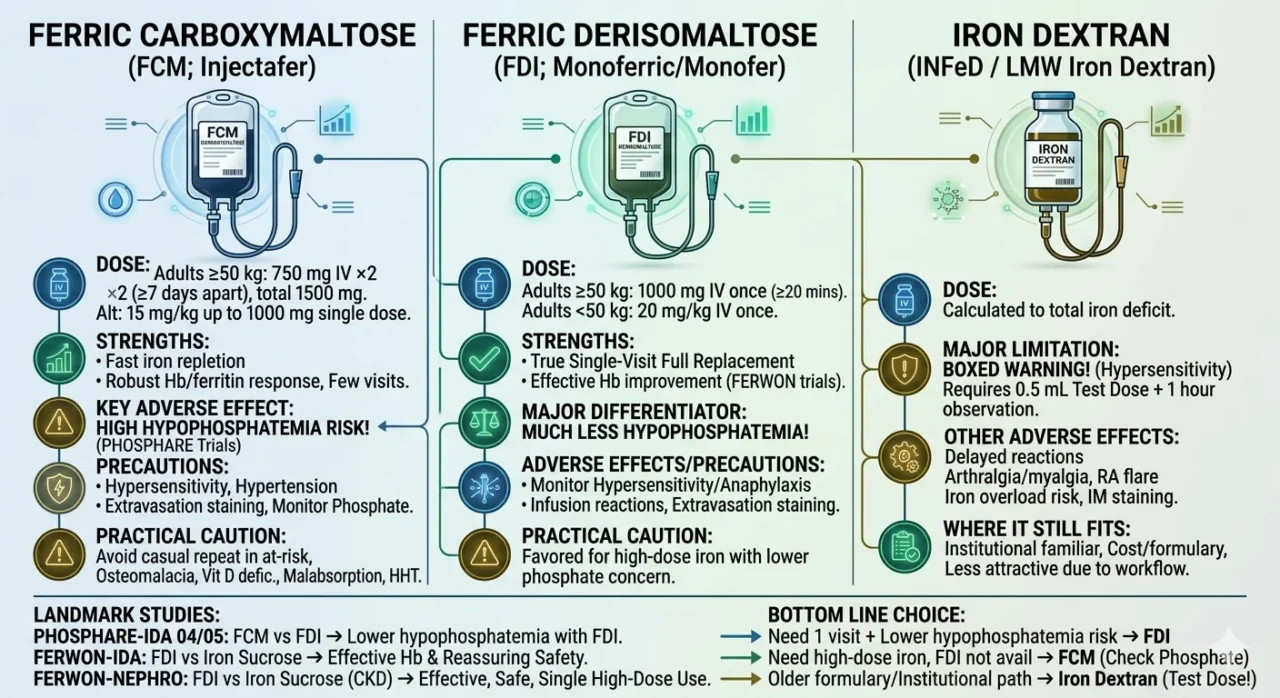
Ferric carboxymaltose (FCM; Injectafer)
- Dose
Adults more than or equal to 50 kg: 750 mg IV ×2 doses at least 7 days apart, total 1500 mg/course; alternative 15 mg/kg up to 1000 mg as a single dose in some adults.
- Strengths and outcomes
Fast iron repletion, robust Hb/ferritin response, widely used when you want high-dose replacement with few visits.
Comparative data support efficacy similar to other modern IV irons.
- Key adverse effect
Biggest issue is hypophosphatemia. In the PHOSPHARE trials, hypophosphatemia was far more common with FCM than with ferric derisomaltose.
- Other precautions
Watch for hypersensitivity, hypertension, extravasation skin staining, and monitor phosphate in at-risk patients or before repeat courses.
Correct pre-existing hypophosphatemia first.
- Practical caution
Avoid casual repeat use in patients with prior FCM-related hypophosphatemia, osteomalacia risk, vitamin D deficiency, malabsorption, HHT, or other phosphate-wasting settings.
Ferric derisomaltose (FDI; Monoferric/Monofer)
- Dose
Adults more than or equal to 50 kg: 1000 mg IV once over at least 20 minutes.
Adults less than 50 kg: 20 mg/kg IV once. Repeat if IDA recurs.
- Strengths and outcomes
Excellent option when you want true single-visit full replacement.
In FERWON-IDA and FERWON-NEPHRO, it showed effective Hb improvement with low rates of serious/severe hypersensitivity, and the pooled safety analysis supported a favorable safety profile.
- Major differentiator
Compared with FCM, FDI causes much less hypophosphatemia.
This is the cleanest evidence-based reason many clinicians prefer it when available.
- Adverse effects and precautions
Still monitor for hypersensitivity/anaphylaxis, infusion reactions, and extravasation with possible long-lasting brown discoloration. Give only where resuscitation capability exists.
- Practical caution
Not ‘zero-risk’ for reactions, but in current practice it is often favored when you want high-dose iron with lower phosphate toxicity concern.
Iron dextran (INFeD / low-molecular-weight iron dextran)
- Dose
Dose is calculated to total iron deficit; the label allows incremental dosing until the calculated total is reached.
- Major limitation
This is the legacy product with boxed-warning hypersensitivity concern.
The U.S. label requires a 0.5 mL test dose before the first therapeutic dose and observation for at least 1 hour before giving the remainder of the first dose. Fatal reactions have occurred even after a tolerated test dose.
- Other adverse effects
Delayed reactions, arthralgia/myalgia, possible flare in rheumatoid arthritis, iron overload risk if diagnosis is wrong, and IM staining issues if given intramuscularly.
- Where it still fits
Useful when institutions are familiar with it or cost/formulary drives use, but from a modern practical standpoint it is usually less attractive than FCM/FDI because of the boxed warning and test-dose workflow.
Bottom line choice
- Need 1 visit and lower hypophosphatemia risk leads to Ferric derisomaltose
- Need effective high-dose iron and FDI not available leads to FCM, but think hard about phosphate
- Using an older formulary option / specific institutional pathway: iron dextran, but respect the boxed warning added test dose added observation
Landmark studies
- Phosphare-ida 04 / phosphare-ida 05: ferric derisomaltose vs ferric carboxymaltose: much lower hypophosphatemia with derisomaltose.
- Ferwon-ida: ferric derisomaltose vs iron sucrose in IDA: effective Hb response and reassuring safety.
- Ferwon-nephro: ferric derisomaltose vs iron sucrose in non-dialysis CKD with IDA: effective and safe, supports single high-dose use.
For iron dextran, modern practice is influenced more by current boxed-warning labeling and older comparative literature than by recent landmark superiority trials.”
Stay updated with Hemostasis Today.
-
Apr 29, 2026, 16:46Deep Vein Thrombosis Risk in Jobs with Prolonged Sitting – NBCA
-
Apr 29, 2026, 16:41Vikramaditya Gangakar: Essential Pharmacy Facts You Should Know About Aspirin
-
Apr 29, 2026, 16:17W. Alberto Sifuentes Giraldo: Non-Criteria Manifestations of Antiphospholipid Syndrome
-
Apr 29, 2026, 16:12Mavis Agnes Kisakye: Concluding a Successful Entrepreneurs Training for Empowering Ptients’ Mothers
-
Apr 29, 2026, 15:48Kriti Batni: Happy to Share on Our Chapter Being Published in Medicine Update 2026
-
Apr 29, 2026, 15:46Dima Shulkin: Comparing HALP and PIV for Mortality Prediction in ACS
-
Apr 29, 2026, 15:44Sonal Sonu: Securing First Prize in Research Oral Presentation at the CME
-
Apr 29, 2026, 15:43Andrew Shaw: Long COVID First Diagnostic Classification with 69% accuracy
-
Apr 29, 2026, 15:41Yazan Abou-Ismail: Contributing Evidence-Based Topic Review on the Evaluation of Easy Bruising

