Aktham El-rekaby: Immunothrombosis in Acute Ischaemic Stroke
Aktham El-rekaby, Clinical Associate Lecturer at Angela Ruskin university, shared a post on LinkedIn:
”Acute Ischaemic Stroke:
Thrombosis with an Immunological Interface
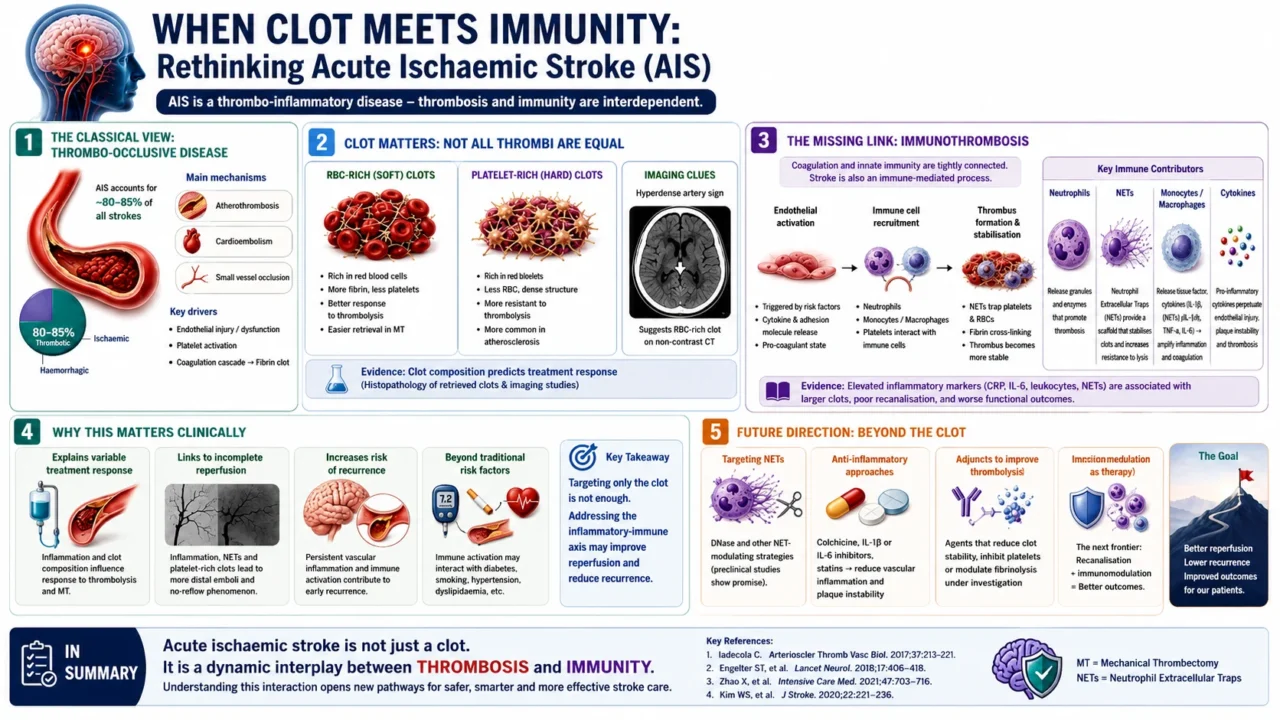
Acute ischaemic stroke (AIS) is fundamentally a thrombo-occlusive disease, most commonly resulting from atherothrombosis, cardioembolism, or small vessel occlusion.
In clinical practice, ischaemic stroke accounts for approximately 80–85percent of all strokes, consistently exceeding haemorrhagic stroke globally, although regional variation exists, with relatively higher haemorrhagic proportions reported in some Asian populations.
Traditionally, the pathophysiology of AIS has been framed around vascular pathology and coagulation dynamics including endothelial injury, platelet activation, and fibrin-rich clot formation.
This paradigm has guided risk stratification and acute therapies such as intravenous thrombolysis and, more recently, mechanical thrombectomy (MT).
Clot Composition and Treatment Response:
Pre-thrombectomy era observations already suggested that clot composition influences treatment efficacy. Thrombolysis is more effective in fibrin-rich (‘soft’) thrombi, whereas platelet-rich or erythrocyte-poor (‘hard’) thrombi tend to be more resistant.
Advances in imaging (e.g., hyperdense artery sign on CT) and histopathological analysis of retrieved thrombi in the MT era have confirmed that:
- Red blood cell (RBC)-rich clots are generally more responsive to thrombolysis and easier to retrieve.
- Platelet-rich clots, often associated with atherosclerosis, are more resistant and structurally compact.
- Clot architecture evolves over time, influenced by platelet activation, fibrin cross-linking, and inflammatory cell infiltration.
The Role of Immunothrombosis
Emerging evidence reframes thrombosis in AIS as part of a broader concept known as immunothrombosis the interaction between the coagulation system and innate immunity.
Key mechanisms include:
- Endothelial activation triggering both coagulation and inflammatory cascades
- Recruitment of neutrophils and monocytes, contributing to clot structure
- Formation of neutrophil extracellular traps (NETs), which stabilize thrombi and increase resistance to thrombolysis
- Release of pro-inflammatory cytokines, amplifying vascular injury and promoting further thrombogenesis.
This suggests that there is an immune-mediated process.
Clinical Implications
Recognizing the immunological contribution to AIS has several implications:
- Explaining variability in treatment response: Differences in clot composition and immune activity may underlie resistance to thrombolysis or incomplete reperfusion with MT.
Risk of recurrence - Persistent inflammatory or immune activation may contribute to ongoing vascular risk beyond traditional factors.
Therapeutic innovation: There is growing interest in immunomodulatory strategies, including:
- Targeting NETs (e.g., DNase-based approaches in experimental models)
- Anti-inflammatory therapies to stabilise plaques and reduce thrombo-inflammation.
- Adjunctive treatments to enhance thrombolysis efficacy.”
Stay updated with Hemostasis Today.
-
May 13, 2026, 15:49Alison Dougall: Oral Health and Cardiovascular Risk in Bleeding Disorders
-
May 13, 2026, 15:38International Journal of Stroke: Oral Anticoagulants and DOAC–Antiplatelet Therapy Are Associated with Increased Major Bleeding Risk
-
May 13, 2026, 15:19Papa Heme։ Causes of Isolated Prolonged aPTT Without Bleeding
-
May 13, 2026, 15:10Wolfgang Miesbach: Rethinking Bleeding Risk in Anticoagulation
-
May 13, 2026, 15:09Veronica Sanchez: Better Cardiovascular Health Helps Lower Heart Attack Risk
-
May 13, 2026, 15:07Tung Nguyen Thanh: New Perspective on Thrombosis in Hematological Malignancies in Vietnam
-
May 13, 2026, 15:03Haji Siyamuddin Ansari: The Key Difference Between Plasma and Serum
-
May 13, 2026, 14:45Masoabi Sefojane: The Cost of a 35-Year Diagnosis Delay
-
May 13, 2026, 14:39Ahmed Mohsen: Managing DVT During Pregnancy and Lactation

