Polycythemia Vera and Thrombosis: Pathogenesis and Prevention
Polycythemia vera (PV), a JAK2-mutated myeloproliferative neoplasm, drives multilineage proliferation through constitutive JAK–STAT activation, creating persistent thrombosis risk affecting 12–39% of patients across all vascular beds.
At the molecular level, PV is most commonly associated with activating mutations in JAK2. These mutations induce constitutive activation of the JAK–STAT signaling pathway, sustaining uncontrolled proliferation.
The JAK2 V617F mutation accounts for the vast majority of cases. Exon 12 mutations define a smaller JAK2-negative subset with similar biological behavior.
PV is not defined by a single driver mutation. Additional cooperating lesions, including TET2, ASXL1, EZH2, and MPL, contribute to disease heterogeneity. They influence phenotype, disease evolution, and thrombotic risk.
Clinically, PV is often detected incidentally on routine blood counts. However, in a significant subset of patients, the first manifestation is vascular. Arterial or venous thrombosis may precede diagnosis, reflecting its systemic nature.
Most cases are sporadic. Rare familial clustering has been reported. This suggests a genetic predisposition in selected families, sometimes with autosomal dominant inheritance patterns.
The major causes of morbidity and mortality in PV are well defined. They include arterial and venous thrombosis, progression to myelofibrosis, and transformation to acute leukemia.
Pathogenesis of Thrombosis in Polycythemia Vera
Why Does Thrombosis Remain The Most Feared Complication in Polycythemia Vera (PV), Even in Treated Patients?
The mechanism reflects a complex interplay between clinical risk factors and intrinsic disease biology. In PV, blood cells are not only quantitatively increased but also qualitatively altered in function.
Vascular risk is elevated across all age groups, including younger patients without prior thrombotic history, and increases further with advanced age or prior thrombosis. The reported incidence of arterial and venous thrombosis in PV ranges from approximately 12–39%.
Thrombotic events are not restricted to the time of diagnosis. They may occur during disease course, including under therapy. This underscores thrombosis as a persistent and biologically driven feature of PV rather than a treatment-neutralized complication.
So Why Does Thrombosis Persist?
The persistence of thrombosis in PV reflects a disease in which multiple components of blood and the vascular system are simultaneously reprogrammed into a sustained prothrombotic state.
Thrombosis in PV affects all vascular beds.
Arterial events (myocardial infarction, stroke, peripheral occlusion) account for 60–70% of complications, while venous thrombosis and unusual sites such as portal vein, mesenteric veins, and Budd–Chiari syndrome are characteristic.
Which Cellular Elements Are Truly Responsible For Driving This Process?
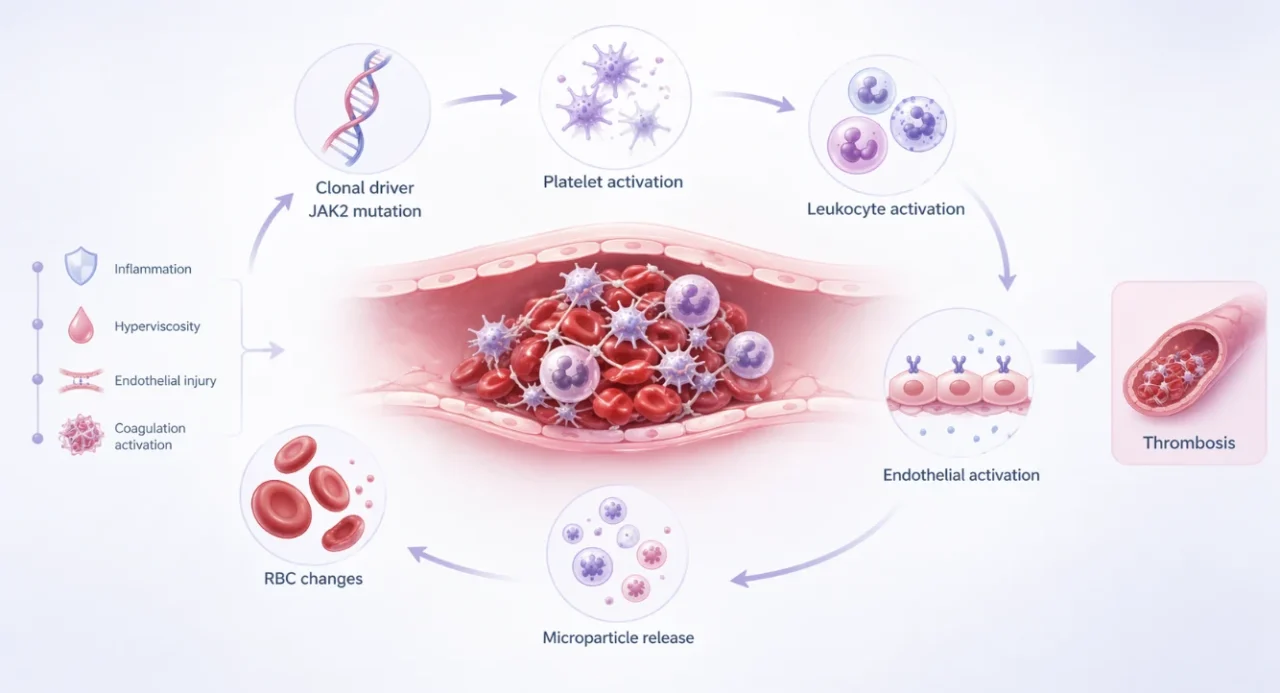
The pathogenesis of thrombosis in polycythemia vera (PV) reflects a sustained disruption of Virchow’s triad, in which blood cells, the endothelium, and rheology are simultaneously altered and amplified by clonal hematopoiesis.
Platelets are central contributors. They exhibit a chronically activated phenotype characterized by increased P-selectin expression, elevated thrombospondin release, and constitutive activation of the GPIIb/IIIa receptor. Rather than resting cellular elements, they function as continuously primed effector cells of thrombosis.
Leukocytes act as inflammatory amplifiers of thrombosis. JAK2 V617F–mutated granulocytes demonstrate upregulation of thrombo-inflammatory pathways, including increased expression of CD11b (αMβ2 integrin), tissue factor, and other leukocyte activation markers.
Neutrophils contribute directly to thrombus formation. They enhance endothelial adhesion and transmigration and release neutrophil extracellular traps (NETs), which promote endothelial injury and thrombin generation.
Activated leukocytes further amplify vascular injury through the release of reactive oxygen species and proteolytic enzymes. This promotes endothelial dysfunction and accelerates thrombin generation.
In this context, inflammation and coagulation are tightly coupled biological processes.
Red blood cells act as active rheological drivers in PV. They are not passive oxygen carriers but become functionally and biomechanically altered.
Increased red cell mass, reduced deformability, and enhanced endothelial adhesion collectively increase blood viscosity and favor microvascular cell–cell interactions.
Endothelium undergoes functional reprogramming toward a prothrombotic surface. This is characterized by increased von Willebrand factor (vWF) release, elevated P-selectin expression, and enhanced platelet adhesion capacity.
In addition, JAK2 V617F mutations may be present within endothelial cells themselves. This results in a directly pro-adhesive and pro-thrombotic vascular phenotype, reinforcing local thrombus formation at the level of the vessel wall.
Integrins represent a key molecular interface in PV-associated thrombosis. They coordinate adhesion between platelets, leukocytes, and the endothelium, sustaining thrombus propagation through continuous cellular cross-talk.
Critical pathways include β1 integrin–VCAM-1 interactions, mediating neutrophil–endothelium adhesion, CD11b (αMβ2 integrin)–driven myeloid activation, and platelet GPIIb/IIIa (αIIbβ3 integrin)–dependent aggregation.
Collectively, these mechanisms establish a multicellular thrombo-inflammatory network.
Platelets, leukocytes, erythrocytes, and endothelial cells act in concert to sustain both thrombus formation and propagation.
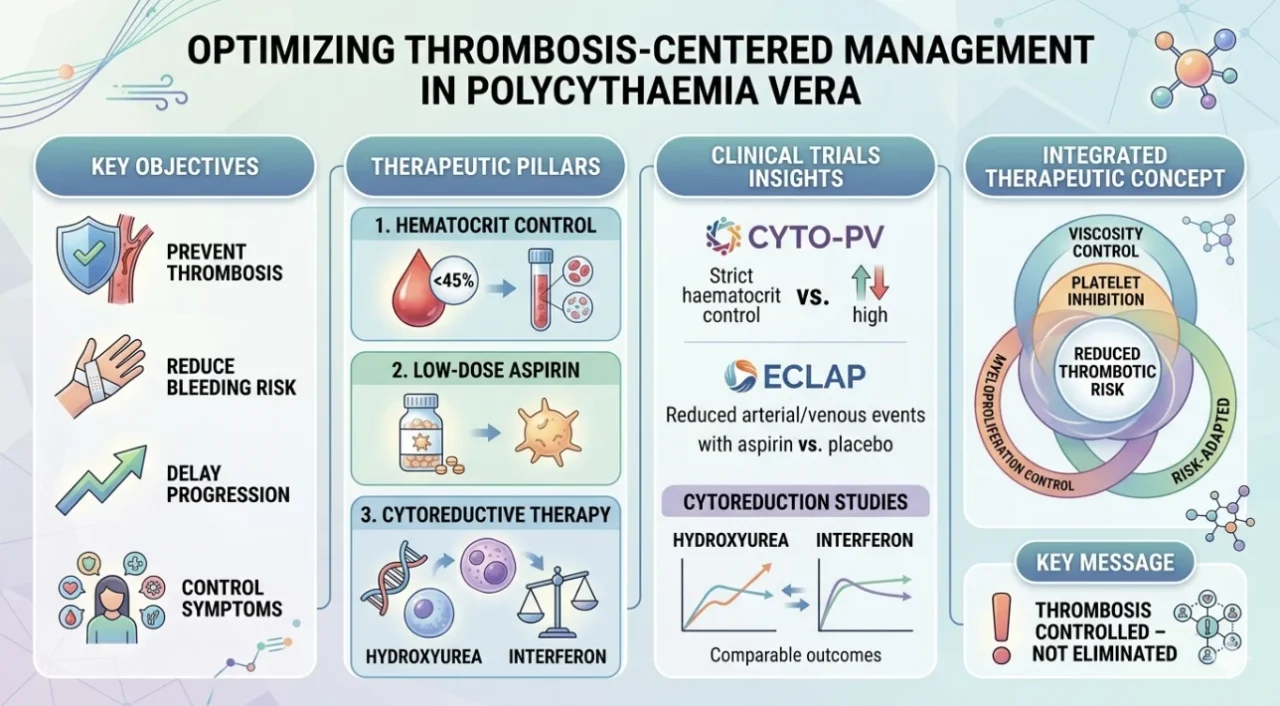
Optimizing Thrombosis-Centered Management in Polycythemia Vera
Clinical Paradigm
In polycythaemia vera (PV), therapeutic success is defined not only by hematologic control but primarily by prevention of thrombotic events, which remain the leading cause of morbidity and mortality.
Despite effective cytoreduction, thrombosis remains the dominant clinical determinant of outcome. Therefore, PV management is fundamentally directed toward long-term suppression of thrombotic risk in a persistent clonal hematopoietic disorder.
Key therapeutic objectives include:
- prevention of arterial and venous thrombosis
- reduction of bleeding risk
- delay of disease progression
- control of symptom burden
1. Hematocrit control: the first evidence-based target
Hematocrit reduction is the first therapeutic axis in PV management and is strongly supported by the CYTO-PV randomized trial.
These findings established hematocrit <45% as the universal treatment target in PV.
However, CYTO-PV also highlighted an important clinical limitation: although hematocrit control significantly reduces risk, it does not abolish thrombosis entirely. This observation underscores that PV-associated thrombosis extends beyond rheologic factors alone.
2. Low-dose aspirin: platelet-directed prevention
Antiplatelet therapy represents the second cornerstone of PV management.
Low-dose aspirin ( 75-81 mg daily ) is generally well tolerated and widely recommended in clinical practice.
The role of aspirin is supported by the ECLAP randomized placebo-controlled trial, which demonstrated that low-dose aspirin significantly reduces arterial and venous thrombotic events by approximately 50–60% in patients with PV, without a meaningful increase in major bleeding. Although no survival advantage was observed, these results firmly established aspirin as a standard component of therapy and a key intervention in guideline-based management.
Bleeding Risk and Acquired Von Willebrand Syndrome: The Paradox of Extreme Thrombocytosis
Bleeding risk in polycythaemia vera represents an important counterbalance to thrombosis prevention, particularly in the setting of extreme thrombocytosis. A key mechanism is acquired von Willebrand syndrome (aVWS), caused by depletion of high-molecular-weight von Willebrand factor multimers due to increased proteolytic cleavage, leading to functional platelet adhesion defects that may not be detected by routine antigen assays.
This risk becomes clinically relevant when platelet counts exceed 1000 × 10⁹/L or in patients with a history of bleeding, where aspirin therapy requires careful individual risk–benefit assessment.
3. Cytoreductive Therapy: Controlling The Disease Driver
Cytoreductive treatment is indicated in high-risk patients and forms the third major pillar of thrombosis prevention.
Hydroxyurea remains the standard first-line agent in older patients, effectively reducing both arterial and venous thrombotic events compared with phlebotomy alone.
In younger patients, interferon-based therapies, including pegylated and ropeginterferon alfa-2b, are increasingly favored due to their ability to induce deeper molecular responses, particularly reduction in JAK2 V617F allele burden.
Ruxolitinib, a JAK1/2 inhibitor used in hydroxyurea-resistant or intolerant patients, has shown benefit in reducing symptom burden and inflammatory activity and is associated with a reduction in thrombotic events in clinical studies, particularly arterial events.
What Do Clinical Trials Tell Us About Thrombosis Prevention?
Evidence from randomized and comparative studies provides a consistent but nuanced picture of cytoreduction in PV.
The MPD-RC 112 trial demonstrated no significant difference in thrombotic outcomes between hydroxyurea and pegylated interferon. Similarly, the PROUD-PV and CONTINUATION-PV studies showed comparable long-term thrombosis rates between interferon- and hydroxyurea-treated patients. These findings suggest that while both strategies are effective, neither demonstrates clear superiority in thrombosis prevention.
In contrast, hydroxyurea has consistently shown superiority over phlebotomy alone in reducing thrombotic complications, confirming the necessity of cytoreduction in high-risk disease. Emerging data with ruxolitinib also suggest meaningful reductions in thrombotic events compared with best available therapy.
Overall, these studies reinforce a key principle: cytoreduction reduces thrombotic risk.
Anticoagulation in Venous Thromboembolism
In patients with established venous thromboembolism, standard anticoagulation with low molecular weight heparin followed by vitamin K antagonists or direct oral anticoagulants reduces recurrence risk by approximately 50%. However, protection remains incomplete.
For unprovoked events, indefinite anticoagulation is generally recommended, provided bleeding risk is acceptable. The decision must always balance persistent thrombotic risk against hemorrhagic complications, particularly in patients receiving concomitant aspirin therapy.
Recurrent Thrombosis
Recurrent thrombotic events remain a major clinical challenge in PV, with an incidence of approximately 7.6 per 100 patient-years. The strongest predictors of recurrence include prior thrombosis, older age, and leukocytosis at baseline or during follow-up. Importantly, recurrent events often mirror the initial thrombotic phenotype, with arterial events tending to recur arterially and venous events recurring in the venous circulation.
A significant proportion of recurrent cases are associated with suboptimal cytoreduction, highlighting the importance of continuous reassessment of treatment adequacy
Are New Drugs Emerging to Prevent Complications in Polycythaemia Vera?
At present, no newly approved therapy has fully replaced hydroxyurea or interferon-based strategies for prevention of thrombotic and other complications in polycythaemia vera (PV). However, the therapeutic landscape is clearly shifting from simple cytoreduction toward disease-modifying approaches targeting the JAK2-driven clone and its inflammatory microenvironment—both central drivers of thrombosis in PV.
Ropeginterferon Alfa-2b: a Step Toward Disease Modification
Among approved therapies, ropeginterferon alfa-2b represents the most important modern advancement.
It demonstrates a progressive increase in complete hematologic response from 32% at 3 months to 63% at 12 months, ensuring sustained hematocrit control and suppression of myeloproliferation—key determinants of thrombotic risk.
Importantly, ropeginterferon also induces reduction in JAK2 V617F allele burden, supporting a potential disease-modifying effect that may translate into long-term reduction of thrombotic events beyond conventional cytoreduction.
Emerging Investigational Strategies
Several next-generation approaches are currently being explored to further reduce disease burden and thrombotic risk:
• JAK2-mutant Selective Inhibitors
Designed to more specifically target the malignant clone, aiming for deeper molecular responses and improved long-term disease control.
• Telomerase Inhibition (e.g., imetelstat)
Targets clonal hematopoiesis at the stem cell level, with potential to impact disease progression and vascular risk biology.
• Combination Strategies (JAK andepigenetic/inflammatory modulation)
Aim to simultaneously suppress clonal proliferation and the inflammatory/endothelial activation that contributes to thrombosis.
While no new agent has yet replaced established first-line therapies, PV management is clearly evolving toward molecularly targeted and disease-modifying strategies rather than isolated hematocrit control.
The therapeutic direction is shifting from simple cytoreduction to durable clonal suppression and modulation of the inflammatory–vascular axis, with the ultimate goal of reducing long-term thrombotic burden.
In polycythaemia vera, the objective is no longer only to control blood counts—but to fundamentally alter the biology driving thrombosis.
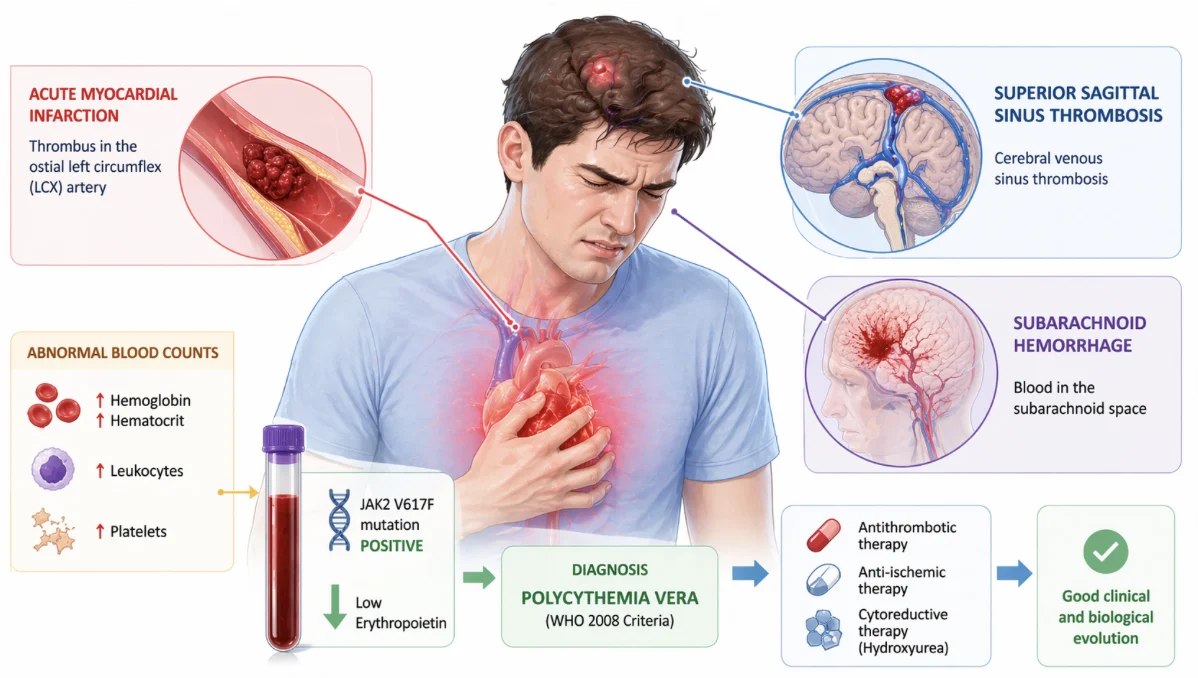
Clinical Case: When Thrombosis Speaks First
A 39-year-old man with no prior cardiovascular history and no identifiable risk factors presents to the emergency department with crushing chest pain and a severe headache persisting for two weeks.
Within hours, the clinical picture becomes extraordinary: STEMI, subarachnoid hemorrhage on CT, and cerebral venous sinus thrombosis on MR angiography.
Arterial thrombosis. Venous thrombosis. Intracranial bleeding.
All in the same patient.
At the same time.
This is not coincidence.
This is biology declaring itself.
Routine laboratory testing reveals the missing pattern: elevated hemoglobin, elevated hematocrit, leukocytosis, and thrombocytosis. Molecular testing then provides the key: JAK2 V617F positivity with low erythropoietin levels, confirming polycythaemia vera.
The diagnosis reframes the entire presentation: in polycythaemia vera, thrombosis may be the opening act, not the finale.
FAQ
1. Why is thrombosis so common in polycythaemia vera?
Thrombosis in PV results from a persistent prothrombotic state caused by increased blood viscosity, activated platelets and leukocytes, endothelial dysfunction, and JAK2-driven inflammatory signaling. This makes thrombosis a core feature of the disease rather than a random complication.
2. Why do patients with PV develop both arterial and venous thrombosis?
PV affects all elements of Virchow’s triad: blood cells, vessel wall biology, and flow dynamics. As a result, patients can develop myocardial infarction, stroke, splanchnic vein thrombosis, deep vein thrombosis, or cerebral venous thrombosis
3. Why can PV patients have bleeding despite high platelet counts?
Extreme thrombocytosis can cause acquired von Willebrand syndrome, which impairs platelet adhesion and increases bleeding risk. This is the main reason aspirin should be used cautiously in patients with very high platelet counts or prior bleeding.
4. What is the role of aspirin in PV?
Low-dose aspirin is a cornerstone of thrombosis prevention in PV because it reduces platelet activation and lowers arterial and microvascular thrombotic risk. Its use must still be balanced against bleeding risk, especially when platelet counts are very high
5. Are new drugs likely to improve thrombosis prevention in PV?
Newer therapies such as ropeginterferon alfa-2b are moving PV management toward deeper disease control, but no agent has fully replaced current standard strategies for thrombosis prevention. The overall direction of care is toward durable suppression of the JAK2-driven clone and its inflammatory vascular effects.
6. Can thrombosis occur even when PV is treated?
Yes. Even with treatment, thrombotic risk is reduced but not eliminated, because the underlying clonal and inflammatory biology persists. This is why long-term risk control remains essential in PV management.
7. What about anticoagulation after venous thrombosis?
Patients with established venous thromboembolism usually require standard anticoagulation, and indefinite treatment is often considered after unprovoked events if bleeding risk allows. The decision should be individualized, especially when aspirin is also being used.
8. When is cytoreductive therapy indicated in PV patients?
Cytoreductive therapy is recommended for high-risk patients, especially those older than 60 years or with prior thrombosis. Its goal is to suppress the disease clone, improve blood count control, and reduce thrombotic complications.
9. Why does thrombosis persist even in treated PV patients?
Multiple reprogrammed elements – activated platelets, NET-releasing leukocytes, rigid RBCs, JAK2+ prothrombotic endothelium – sustain multicellular thrombo-inflammation beyond cytoreduction alone.
10. What is the most important target for thrombosis prevention?
The best-established target is hematocrit below 45%. This threshold is associated with fewer cardiovascular deaths and major thrombotic events compared with less strict control.
Written by Elen Avetisyan, MD
Stay updated with Hemostasis Today.
-
May 11, 2026, 01:38Heghine Khachatryan: The Emerging Era of Immunothrombosis and Precision Vascular Medicine
-
May 10, 2026, 18:07Recurrent DVT and PE in Young Adults May Indicate an Underlying Cause – The Lancet Group
-
May 10, 2026, 18:06Noemi Zampatti: Pediatric Celiac Crisis Complicated by Refeeding Syndrome and Proposed Diagnostic Criteria
-
May 10, 2026, 18:05Giovanni Solimeno: When the Common Femoral Artery Wall is Diseased, Closure is Not a Step
-
May 10, 2026, 18:04Maitri Vaishnav: The Bombay Blood Group and the Critical Role of the H Antigen
-
May 10, 2026, 18:03Nicolas Hubacz: Mesenchymal Stem Cells Are Some of the Most Fascinating Cells in Regenerative Medicine
-
May 10, 2026, 16:20Joshua Ngimbwa: How Sex and Socioeconomic Factors Influence Stroke Outcomes in Tanzania
-
May 10, 2026, 16:18Gustavo Monnerat: How Thousands of Fake References Slipped Into Biomedical Literature
-
May 10, 2026, 16:13Dalius Jatužis: Our AI-Driven Automated Stroke Registry Data Extraction Gets Best Poster Award at ESOC 2026

