
Khaled Musallam Shares BEYOND Trial Phase 2 Insights on Luspatercept Use in NTD β-Thalassaemia
Khaled Musallam, Group Chief Research Officer at Burjeel Holdings, shared on LinkedIn:
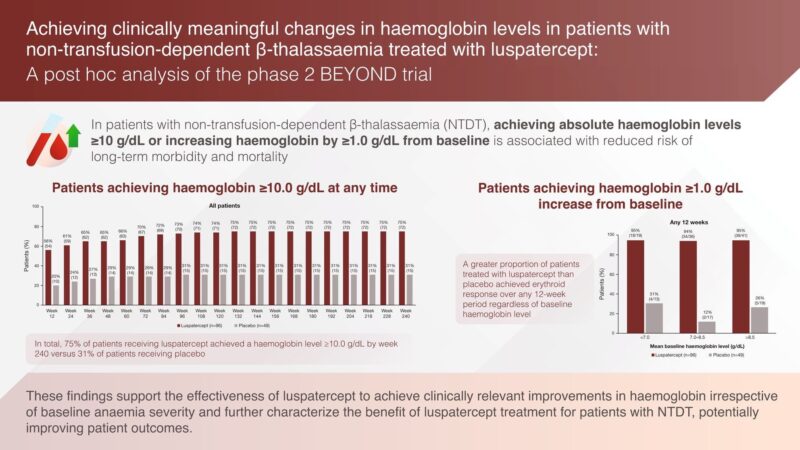
”Pleased to share our new paper out today in the British Journal of Haematology.
This is a post-hoc analysis of the BEYOND trial specifically looking at the proportion of NTDT patients that reached and sustained a hemoglobin level of >10 g/dL while on ß, reflecting clinically meaningful impact considering this would now place them in the ‘safe zone’.”
Read the full article in BJHaem.
Article: Achieving clinically meaningful changes in haemoglobin levels in patients with non-transfusion-dependent β-thalassaemia treated with luspatercept: A post hoc analysis of the phase 2 BEYOND trial
Authors: Khaled Musallam X, Ali T. Taher, John B. Porter, Antonis Kattamis, Mrudula B. Glassberg, Luciana Moro Bueno, Patricia Martin-Regueira, Marta Reverte, Loyse Felber Medlin, Matthew Dyer, Kefeng Wang, Maria Domenica Cappellini
Stay updated on all scientific advances with Hemostasis Today.
-
Apr 30, 2026, 08:11Navya Sree: Breaking Down DVT From Theory to Practice
-
Apr 30, 2026, 06:06Shashank Joshi: The Emerging Role of Inflammatory Biomarkers in Acute CVS Thrombosis
-
Apr 30, 2026, 05:30John Bramble: Ensuring Safe AI Integration in Medicine
-
Apr 30, 2026, 04:55Danielle Stull: Raising Awareness of CVS Thrombosis in Young Women
-
Apr 29, 2026, 17:25Robert Brodsky: Ravulizumab Appears To Be Safe And Effective In Pregnancy
-
Apr 29, 2026, 16:53Chokri Ben Lamine: IV Iron Showdown – FCM vs Ferric Derisomaltose vs Iron Dextran
-
Apr 29, 2026, 16:46Deep Vein Thrombosis Risk in Jobs with Prolonged Sitting – NBCA
-
Apr 29, 2026, 16:41Vikramaditya Gangakar: Essential Pharmacy Facts You Should Know About Aspirin
-
Apr 29, 2026, 16:17W. Alberto Sifuentes Giraldo: Non-Criteria Manifestations of Antiphospholipid Syndrome

