
Vikas Londhe on Andexxa’s Safety Assessment Updates
Vikas Londhe, Chief Editor of Pharmacally.com, shared on LinkedIn:
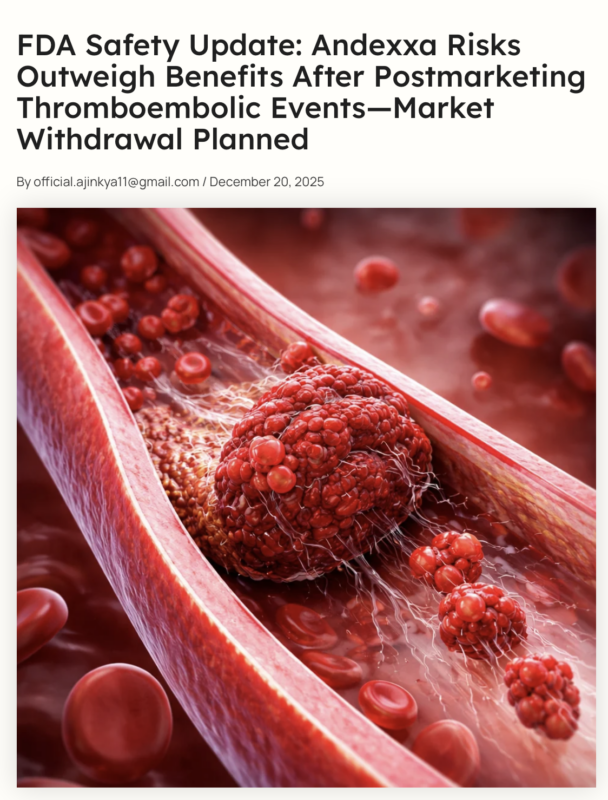
”FDA Safety Update: Andexxa Risks Outweigh Benefits After Postmarketing Thromboembolic Events—Market Withdrawal Planned
FDA Updates Safety Assessment of Andexxa After Serious Thromboembolic Events.
On December 18, 2025, the U.S. Food and Drug Administration (FDA) issued a safety communication regarding Andexxa (coagulation factor Xa (recombinant), inactivated-zhzo).
The agency has reviewed postmarketing safety data showing an increased incidence of thromboembolic events, including serious and fatal outcomes, in patients treated with Andexxa.
Based on these findings, the FDA now considers that the risks associated with Andexxa outweigh its benefits in its approved use.”
Read the full announcement here.
Stay updated with Hemostasis Today.
-
May 25, 2026, 05:25Abdullah Mohsin: Automated Detection of DVT Using Deep Learning and Doppler Ultrasound
-
May 25, 2026, 05:07The Dual Role of PAI-1 Mutations in Bleeding and Thrombotic Disorders – ICHCC
-
May 25, 2026, 05:04Fibrinogen αC Domain Lysines Drive Plasminogen Binding and Plasmin Generation – JTH
-
May 25, 2026, 04:50Gülşah Türkoğlu: An Encouraging Momentum Around the Adoption of Resolutions on Stroke Care at WHA79
-
May 25, 2026, 04:47Krishnakant Prasad: A Quick Clinical Review of Renal Papillary Necrosis
-
May 25, 2026, 04:40Rajiv Kumar Jain: World Health Assembly Adopts First-Ever Global Resolution on Stroke Prevention and Care
-
May 25, 2026, 04:23Sarfraz Ahmad: Diagnostic and Therapeutic Challenges in ICA Aneurysm Presenting With Epistaxis
-
May 24, 2026, 17:48Flora Peyvandi: Crucial Role of Placental ADAMTS13 in Perinatal Survival
-
May 24, 2026, 17:13Mahtab Mashayekhi: The Evolving Role of Red Blood Cell Exchange in the Management of SCD

