
FDA Approves Jakafi XR (Ruxolitinib Extended-Releas) for Myelofibrosis, Polycythemia Vera and GVHD
Jakafi XR (ruxolitinib extended-release tablets) received U.S. Food and Drug Administration approval for the treatment of Myelofibrosis (MF), Polycythemia Vera (PV), and Graft-versus-host disease (GVHD).
In May 2026, Incyte announced this approval of Jakafi XR, outlining the following indications:
- adults with intermediate- or high-risk MF
- adults with PV who are resistant or intolerant to hydroxyurea
- adults and children aged 12 years and older with steroid-refractory acute GVHD
- adults and children aged 12 years and older with chronic GVHD after failure of one or two prior lines of systemic therapy
About PV, MF and GVHD
Myelofibrosis and Polycythemia Vera are myeloproliferative neoplasms. The majority of patients harbor a somatic mutation in one of three genes involved in the JAK-STAT pathway: JAK2, CALR, or MPL.
PV is characterized by clonal overproduction of red blood cells and, in some cases, leukocytes and platelets. The resulting increase in blood viscosity and thrombotic tendency raise the risk of complications such as deep vein thrombosis, stroke, and myocardial infarction. Common symptoms include headache, pruritus, erythromelalgia and splenomegaly.
MF is characterized by clonal proliferation of malignant hematopoietic stem and progenitor cells, aberrant cytokine production, and progressive bone marrow fibrosis, which can lead to severe anemia, fatigue, and hepatosplenomegaly. MF may arise de novo as primary myelofibrosis (PMF) or develop secondarily from Essential Thrombocythemia or Polycythemia Vera.
GVHD is a serious immune-mediated complication of allogeneic hematopoietic stem cell transplantation in which donor immune cells attack recipient tissues. There are two major forms of GVHD: acute, which generally occurs within 100 days of transplant, and chronic, which generally occurs after 100 days of transplant. Both forms can affect multiple organ systems, including the skin, gastrointestinal tract and liver. GVHD remains a major cause of post-transplant morbidity, mortality and impaired quality of life.
Ruxolitinib: Evolution of Approved Indications
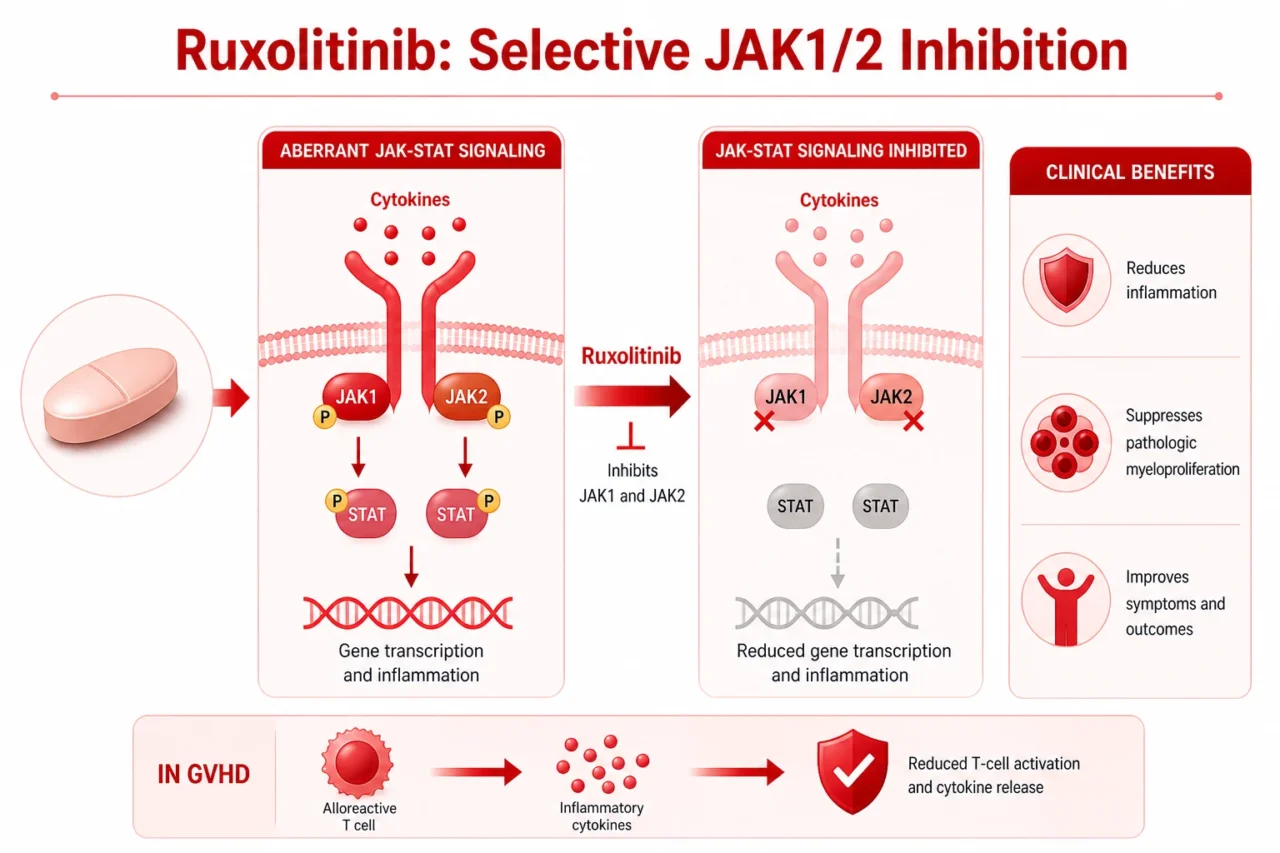
Ruxolitinib is an oral selective inhibitor of Janus kinase 1 (JAK1) and JAK2. By inhibiting aberrant JAK-STAT signaling, it reduces cytokine-driven inflammation, suppresses pathologic myeloproliferation, and improves disease-related symptoms. In GVHD, JAK inhibition also modulates alloreactive T-cell signaling and inflammatory cytokine release.
Ruxolitinib received its first FDA approval in 2011 for intermediate- or high-risk MF, based on the phase 3 COMFORT-I and COMFORT-II trials, in which ≥35% spleen volume reduction was achieved in 42% vs <1% with placebo at week 24 and 28% vs 0% with best available therapy at week 48, respectively.
In 2014, the FDA approved ruxolitinib for PV resistant or intolerant to hydroxyurea, supported by the phase 3 RESPONSE trial, in which hematocrit control without phlebotomy was achieved in 60% vs 20% with best available therapy.
In 2019, ruxolitinib was approved for steroid-refractory acute GVHD based on the phase 2 REACH1 trial, demonstrating an OS rate of 57% and CR rate of 31% at day 28. It was later established as the standard second-line therapy through the phase 3 REACH2 trial.
In 2021, the FDA approved ruxolitinib for chronic GVHD after failure of one or two prior systemic therapies, supported by the phase 3 REACH3 trial, where the OS rate was 70% vs 57% with best available therapy.
What Is Jakafi XR?
Jakafi XR is a once-daily, film-coated, extended-release formulation of Jakafi (ruxolitinib).
FDA approval was based on a clinical study which demonstrated that a single 55 mg Jakafi XR tablet taken once-daily (QD) is bioequivalent to a single 25 mg Jakafi tablet, the immediate-release (IR) dosage form, taken twice-daily (BID). This means that, based on key measures of steady-state drug exposure, it delivers the same active ingredient at comparable levels throughout the day, indicating the potential for similar clinical benefit.
Safety Profile
The safety of Jakafi XR has been established from adequate and well-controlled studies. The most common side effects of JAKAFI or JAKAFI XR include:
- For certain patients with MF and PV: low platelet or red blood cell counts, bruising, dizziness, headache and diarrhea.
- For patients with acute GVHD: low platelet counts, low red or white blood cell counts, infections and swelling.
- For patients with chronic GVHD: low red blood cell or platelet counts and infections, including viral infections.
Why This Approval Matters
The approval of Jakafi XR is meaningful for patients with chronic conditions such as MPNs and GVHD, who often face complex long-term treatment regimens and multiple comorbidities. A once-daily formulation may simplify therapy, improve convenience and provide an additional dosing option for eligible patients. Not every advance is a new drug, sometimes it is a better way to deliver one.
More details are available here.
Stay Informed with Hemostasis Today.
-
May 5, 2026, 16:28Nikolay Novitski: A New Era in Cardiovascular Prevention
-
May 5, 2026, 16:23Satyam Arora: Best Abstract Award in Pediatric Apheresis at ASFA 2026 Congress
-
May 5, 2026, 16:17Hannah Omunakwe: Your Sick Child Has a Clot. Did You See That Coming?
-
May 5, 2026, 15:57Venous Thromboembolism Risk in Pregnancy and the Postpartum Period – NBCA
-
May 5, 2026, 15:28Soumen Bhattacharyya: Hyperhomocysteinemia and Thrombosis – What Clinicians Should Know
-
May 5, 2026, 15:06Wolfgang Miesbach: How Should We Approach GI Angiodysplasia in VWD?
-
May 5, 2026, 14:48What Constitutes High Risk for Venous Thromboembolism? – JTH
-
May 5, 2026, 14:41How Real-Time Data, Rehabilitation Registries, and Patient-Reported Outcomes Can Transform Practice – European Stroke Organisation
-
May 5, 2026, 14:41Driving National Change for Bleeding Disorders – WFH

