
66-Country Survey Exposes Barriers to Hemophilia Gene Therapy
Wolfgang Miesbach, Professor of Medicine at Frankfurt University Hospital, posted on LinkedIn:
”Haemophilia Gene Therapy: New ISTH survey reveals a critical disconnect between clinical innovation and healthcare readiness.
Global survey assessing knowledge, perceptions, and clinical preparedness for gene therapy among haemophilia professionals. Over eight months (August 2023–January 2024), 327 healthcare professionals from 66 countries responded via an anonymous online questionnaire distributed through four major professional organizations.
Current Therapy Limitations
– burden of dosing schedules and prophylaxis
– of inhibitor development
– with injections and venous access
– and reimbursement pressures
– with treatment adherence
– impact on quality of life and joint disease
– breakthrough bleeding and pain
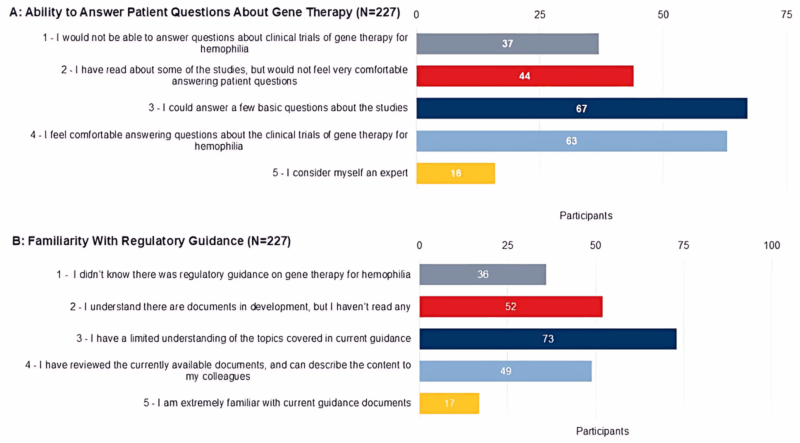
What Was Found Related to Gene Therapy
– 7.5% feel extremely familiar with gene therapy
– 35.6% uncomfortable answering patient questions about trials
– 60% of patients have never discussed gene therapy with their care team
Major Barriers to Implementation of Gene Therapy
– Durability uncertainty
– Variability in patient response
– Cost and reimbursement challenges
– Restrictive eligibility criteria
– Insufficient healthcare system preparedness
Gene therapy is clinically available. What’s missing is confidence, system readiness, and equitable global access. Closing this gap requires targeted professional education, healthcare pathways, and solutions that ensure all eligible patients – ideally, regardless of geography – can access this transformative treatment.
Published in Therapeutic Advances in Hematology, open access link ”
Title: Gene therapy for hemophilia: results of ISTH global survey on current knowledge, attitudes, and preparedness of the hemophilia care team
Authors: Melissa F. Glasner, Wolfgang Miesbach
Stay informed with Hemostasis Today.
-
May 29, 2026, 13:39Kalyan Roy: Hematopoietic Stem Cell Transplantation in Thalassaemia
-
May 29, 2026, 12:53Wolfgang Miesbach: The Risk of Deskilling in Modern AI Assisted Medicine
-
May 29, 2026, 12:20Pall T. Onundarson: Is Anonymous Peer Review Always Objective
-
May 29, 2026, 11:59Jolene Ng: New Review Explores the Pathophysiology of Pre-Eclampsia
-
May 29, 2026, 11:44Lynne Baird: Why Expanding Bleed Control Kits Across Public Transport Matters
-
May 29, 2026, 11:30Rumbidzai Mushambi: When ‘Just Hormones’ Delays Real Diagnosis
-
May 29, 2026, 09:53Yassin Taher: Recognizing Clonal Eosinophilic Disorders in Pediatric Hypereosinophilic Syndrome
-
May 29, 2026, 09:52Sergio Emanuel Kaiser: Ruvonoflast and a New Anti-Inflammatory Strategy for Residual Cardiovascular Risk
-
May 29, 2026, 09:50Maria Symiakaki: Advancing Cardiovascular Prevention at the 2026 Congress of the European Atherosclerosis Society

