
Ajay Kumar: Plasma Component Quality Control Standards
Ajay Kumar, Senior Resident at Geetanjali Hospital, shared a post on LinkedIn:
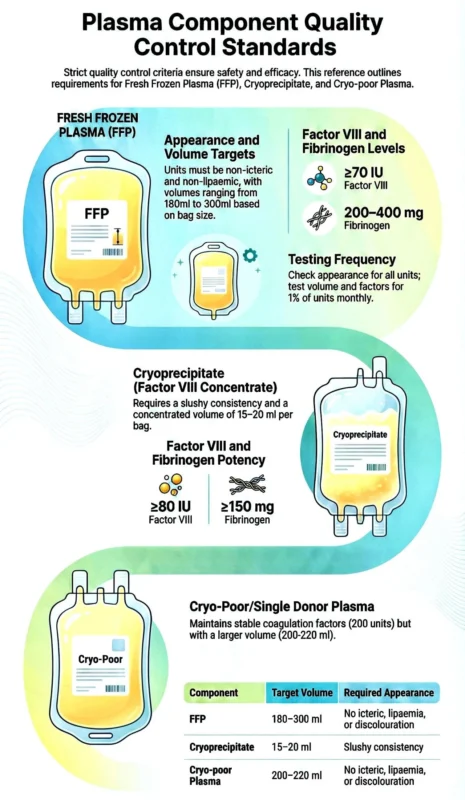
“Blood centres are not just collection units, they are highly regulated manufacturing facilities operating under strict quality frameworks.
Different plasma components such as Fresh Frozen Plasma (FFP), Cryoprecipitate, and Cryo Poor Plasma are prepared through standardized processes that require precision, validated equipment, trained personnel, and robust documentation.
Each of these components must meet stringent quality control criteria as mandated by Directorate General of Health Services (DGHS) guidelines and Drugs and Cosmetics Act, 1940 and Rules, 1945.
Quality in transfusion medicine is not optional. It is regulatory, ethical, and lifesaving.”
Stay updated with Hemostasis Today.
-
May 31, 2026, 16:29José Antonio García Erce: Collaborative PBM Implementation in Transfusion Centers and Services
-
May 31, 2026, 16:22Vera Ignjatović: How Do We Rigorously Study These Drugs in Children to Understand Their True Benefits and Risks?
-
May 31, 2026, 16:21Ksenija Stach: Is This The Future of Lipid-Lowering Lherapy?
-
May 31, 2026, 15:42Antonio Greco: Balancing Bleeding and Thrombotic Risk After PCI
-
May 31, 2026, 15:41Eric Topol: Replication of Plasma p-tau217 Blood Test for Predicting Alzheimer’s Disease
-
May 31, 2026, 15:32ASH Invites Dynamic Members to Apply for 2027 Leadership and Committee Roles
-
May 31, 2026, 14:39Monique Gore-Massy: Redefining Lupus Innovation Through Patient Partnership
-
May 31, 2026, 14:31Mirjam Teboe: The Golden Rule Behind Reliable Lupus Anticoagulant Testing
-
May 31, 2026, 14:20Improving Access to Care for People Living with Haemophilia and Haemoglobinopathies – NNHF

