
Ifeanyichukwu Ifechidere: What If Altuvoct Redefined a Biological Limit in Hemophilia A?
Ifeanyichukwu Ifechidere, Biomedical Scientist at Sheffield Teaching Hospitals NHS Foundation Trust, shared a post on LinkedIn:
“What If Haemophilia A Just Broke One of Its Biggest Biological Limits with the New Treatment Altuvoct?
Introduction
Haemophilia A is a genetic bleeding disorder caused by deficiency or dysfunction of Factor VIII, a very important protein in the blood coagulation cascade.
When there is inadequate Factor VIII (FVIII), patients are unable to form stable blood clots, which leads to prolonged bleeding after injury and spontaneous internal bleeding episodes, particularly in joints and muscles.
For decades, clinical treatment plans and strategies have focused on replacing the missing factor using recombinant FVIII therapies.
Although recombinant technology has revolutionised haemophilia care, one persistent biological constraint has limited the effectiveness of these therapies: the short half-life of Factor VIII in circulation.
Most FVIII molecules survive in the bloodstream for only about 12–19 hours, meaning patients often require frequent infusions to maintain protective levels.[3]
The reason behind this limitation lies in a physiological partnership between FVIII and another protein, von Willebrand factor (VWF).
In the bloodstream, more than 95% of circulating FVIII is bound to VWF, which stabilises it but also determines how quickly it is cleared from circulation.
When VWF is removed from the blood, FVIII disappears with it.
This phenomenon is commonly described as the ‘VWF half-life ceiling.'[3]
A new therapy—Altuvoct (efanesoctocog alfa)—has been engineered specifically to overcome this biological limit.
The drug represents a new generation of bioengineered clotting factor therapy designed to extend the survival of FVIII in the bloodstream and potentially transform haemophilia A management.
This article explores how Altuvoct works, its molecular design, and why many scientists believe it may represent a major breakthrough in haemophilia treatment.
Understanding the Biological Limit: The VWF Ceiling
To understand the significance of Altuvoct, it is important to first examine why FVIII therapies have historically struggled to achieve long half-lives.
FVIII is a large glycoprotein that plays a key role in the intrinsic pathway of the coagulation cascade. It acts as a cofactor for Factor IXa, helping convert Factor X to Factor Xa, which ultimately leads to thrombin generation and fibrin clot formation.[5]
However, FVIII is unstable when circulating freely. In healthy individuals, it is protected by binding to VWF, a large multimeric protein that acts as a carrier molecule.
While this interaction stabilises FVIII, it also creates a limitation: the lifespan of FVIII becomes dependent on the clearance rate of VWF.
As a result, even advanced recombinant FVIII therapies—including extended half-life (EHL) products—remain partially constrained by this biological partnership.
The clearance of the FVIII-VWF complex effectively places a physiological ceiling on how long FVIII can remain active in the bloodstream.[9]
For patients, this means that even modern FVIII therapies often require intravenous administration multiple times per week to maintain protective clotting levels.
Introducing Altuvoct: A New Generation of FVIII Therapy
Altuvoct (efanesoctocog alfa) is a recombinant fusion protein designed to break through the VWF half-life ceiling.
It represents a new class of FVIII replacement therapy that combines multiple bioengineering strategies into a single molecule.[1]
Unlike conventional FVIII products, Altuvoct has been engineered to circulate independently of endogenous VWF, allowing other half-life extension technologies to function more effectively.[9]
This design has produced remarkable pharmacokinetic results. Studies show that efanesoctocog alfa can achieve a half-life approximately three to four times longer than standard FVIII products, allowing many patients to maintain protective FVIII levels with once-weekly dosing.[1]
The key to this innovation lies in the drug’s unique molecular architecture.
The Molecular Structure of Altuvoct
Altuvoct is not simply a modified FVIII molecule—it is a complex fusion protein combining several engineered components that work together to extend its lifespan in the bloodstream.
Core components of the molecule
The drug contains four major structural elements:
- B-domain deleted recombinant FVIII
- Human IgG1 Fc fragment
- VWF D’D3 domain
- Two XTEN polypeptide chains
Together, these components form a heterodimeric fusion protein designed to maximise stability and prolong circulation time.[5]
Simplified molecular diagram
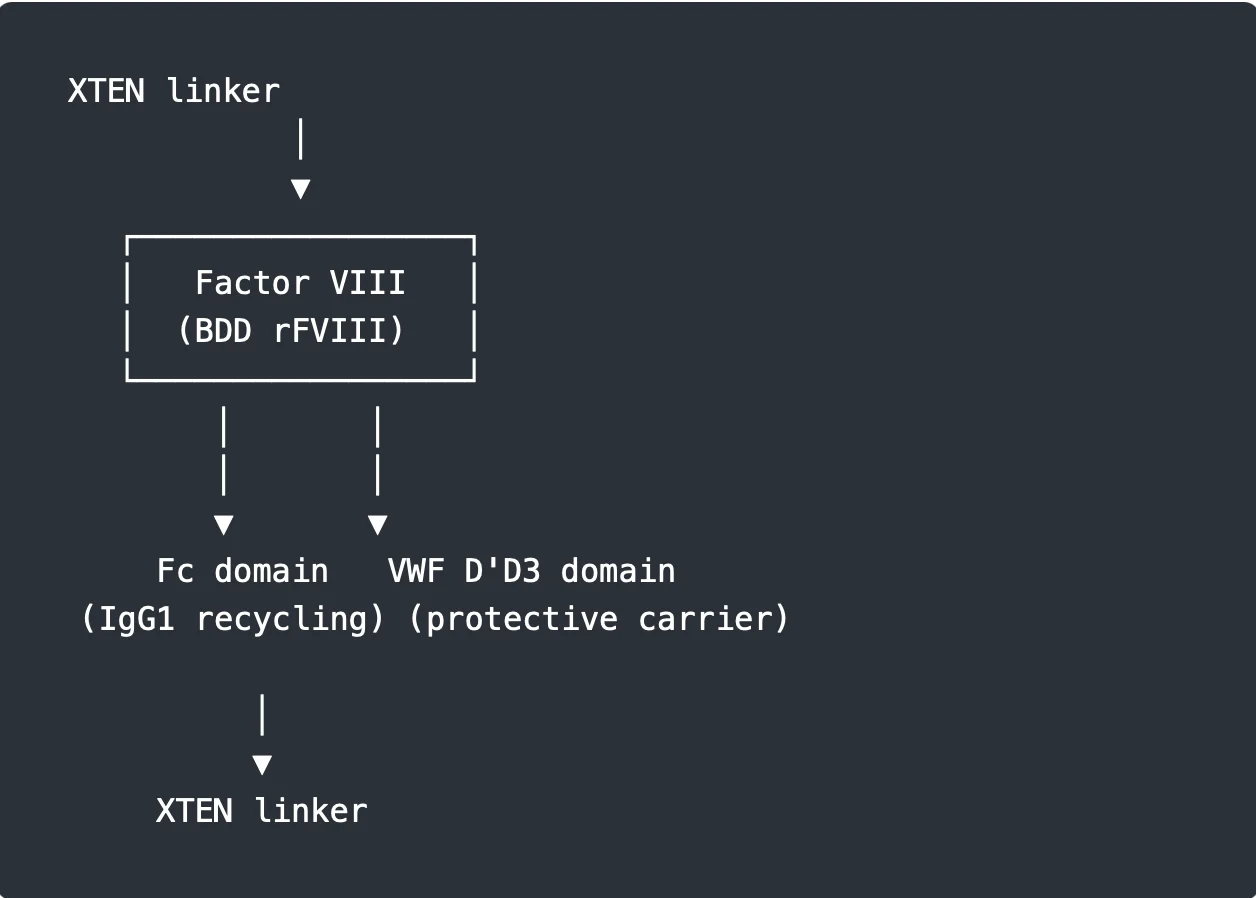
Each component serves a specific functional role in overcoming FVIII clearance.
Breaking the VWF Ceiling: How the Molecule Works
The most innovative feature of Altuvoct is the incorporation of the D’D3 domain of VWF directly into the drug molecule.
1. Preventing binding to endogenous VWF
The D’D3 domain is the portion of VWF responsible for binding FVIII.
By attaching this domain directly to the recombinant FVIII molecule, the drug effectively occupies its own binding site.
This prevents the molecule from attaching to endogenous VWF circulating in the patient’s blood.
As a result, the drug is no longer cleared according to the natural VWF turnover rate, thereby bypassing the traditional half-life ceiling.[9]
2. Fc-mediated recycling
The second key innovation is the addition of an IgG1 Fc fragment.
This region interacts with the neonatal Fc receptor (FcRn), a cellular recycling system that rescues proteins from lysosomal degradation and returns them to circulation.
Many long-acting biologic drugs use this mechanism to extend their half-life.
In Altuvoct, FcRn recycling helps the molecule persist in the bloodstream for longer periods.[7]
3. XTEN technology
The molecule also contains two XTEN polypeptides, long chains of hydrophilic amino acids that increase the molecule’s size and hydrodynamic radius.
These flexible polymers act as protective shields that reduce renal filtration and proteolytic degradation, further extending the drug’s pharmacokinetic profile.[4]
Combined effect
Together, these modifications allow Altuvoct to remain active in the bloodstream far longer than traditional FVIII therapies.
Pharmacokinetics and Clinical Impact
The molecular design of Altuvoct translates directly into improved pharmacokinetic performance.
Clinical studies show that efanesoctocog alfa can achieve:
- Half-life of approximately 40–50 hours
- Sustained FVIII activity in the near-normal range for several days
- Protective clotting levels maintained for up to one week after infusion.[7]
This means that many patients may require only one infusion per week, significantly reducing treatment burden compared with earlier FVIII therapies that required multiple infusions each week.
Example patient scenario
Consider a patient with severe haemophilia A receiving standard FVIII therapy:
- Infusions every 2–3 days
- FVIII levels dropping rapidly between doses
- Increased risk of bleeding during trough periods
With Altuvoct:
- Once-weekly dosing
- Sustained FVIII levels throughout the week
- Reduced fluctuations in clotting protection
Such improvements could significantly enhance quality of life and treatment adherence.
Why This Innovation Matters
The development of Altuvoct represents a shift in how scientists approach haemophilia treatment.
Instead of simply replacing missing FVIII, researchers have re-engineered the molecule itself to overcome a fundamental biological limitation.
This innovation reflects broader trends in modern biomedical science:
- Protein engineering
- Fusion protein therapeutics
- Pharmacokinetic optimisation
By combining multiple technologies—Fc fusion, VWF domain engineering, and XTEN linkers—scientists have created a molecule that behaves very differently from conventional clotting factors.
Some researchers have described this as a ‘renaissance of Factor VIII replacement therapy.’ [1]
The Future of Haemophilia Therapy
While Altuvoct is a significant advance, it is part of a rapidly evolving therapeutic landscape.
Other emerging strategies for haemophilia A include:
- Bispecific antibodies, such as Emicizumab, which mimic FVIII activity
- Gene therapy approaches
- RNA-based therapies
Each of these treatments offers different advantages. Some experts believe the future may involve combining multiple therapeutic strategies rather than relying on a single approach.
However, for patients who prefer or require FVIII replacement therapy, Altuvoct may represent one of the most sophisticated versions of that strategy to date.
Conclusion
Haemophilia A treatment has evolved dramatically over the past several decades—from plasma-derived factors to recombinant proteins and now to advanced bioengineered fusion molecules.
Altuvoct represents a major milestone in this journey. By breaking the long-standing VWF half-life ceiling, the drug demonstrates how molecular engineering can overcome fundamental biological constraints.
Through the combination of:
- VWF domain engineering
- Fc receptor recycling technology
- XTEN polymer shielding
scientists have created a molecule capable of circulating independently of endogenous VWF and remaining active for significantly longer periods.
The result is a therapy that may reduce treatment burden, maintain higher clotting factor levels, and potentially improve outcomes for people living with haemophilia A.
While long-term data and real-world experience will continue to shape our understanding of its role in treatment, one thing is clear: Altuvoct illustrates how modern biotechnology can reshape the boundaries of what is biologically possible in medicine.
References
- Dargaud Y, et al. Efanesoctocog alfa: the renaissance of Factor VIII replacement therapy. Haematologica. 2024.
- Konkle BA, et al. Efanesoctocog alfa prophylaxis for hemophilia A. New England Journal of Medicine.
- Demers M, et al. Efanesoctocog alfa elicits functional clot formation that is indistinguishable to that of recombinant factor VIII.
- Springer Nature. Efanesoctocog alfa in hemophilia A: a profile of its use.
- National Cancer Institute Drug Dictionary. Efanesoctocog alfa.
- Springer Nature. Efanesoctocog alfa: first approval.
- Clinical pharmacology reports on efanesoctocog alfa mechanism and half-life extension.
- Research on FVIII-VWF interaction and biological half-life limitation.
- Efanesoctocog Alfa: A Comprehensive Clinical and Pharmacological Analysis of a First-in-Class Factor VIII Replacement Therapy“
-
May 6, 2026, 02:58Limiaa Babikir: What Is Polyspecific DAT?
-
May 6, 2026, 02:47Karen Rollinson: Grateful to Be Connecting with so Many Families and Advocates at HFA 2026 in New Orleans
-
May 6, 2026, 02:38Moustafa Abdou: Is the Bone Marrow Examination Still the ‘Gold Standard’?
-
May 6, 2026, 02:23Khaled Musallam: When the Worlds of Thalassemia and Hemophilia Converge
-
May 6, 2026, 02:12Caitlin Raymond: How STOP-BABESIOSIS Just Rewrote 40 Years of Practice
-
May 5, 2026, 16:28Nikolay Novitski: A New Era in Cardiovascular Prevention
-
May 5, 2026, 16:23Satyam Arora: Best Abstract Award in Pediatric Apheresis at ASFA 2026 Congress
-
May 5, 2026, 16:17Hannah Omunakwe: Your Sick Child Has a Clot. Did You See That Coming?
-
May 5, 2026, 15:57Venous Thromboembolism Risk in Pregnancy and the Postpartum Period – NBCA

