
Wolfgang Miesbach Shares Newly Released Gene Therapy Qualification Criteria Paper
Wolfgang Miesbach, Professor of Medicine at Frankfurt University Hospital, shared a post on X:
“Exciting News! Our opinion paper on gene therapy qualification criteria has just been published! It outlines comprehensive standards for hub-and-spoke centers delivering gene therapy for haemophilia, helping ensure safety and efficacy across Europe and beyond:
Timely infrastructure guidance: The paper establishes practical qualification criteria for haemophilia centres delivering gene therapy, helping address new organizational, technical, and clinical requirements as gene therapies move into real-world practice.
Expert consensus: A comprehensive survey achieved 100% response among interdisciplinary specialists, ensuring robust recommendations grounded in broad clinical experience.
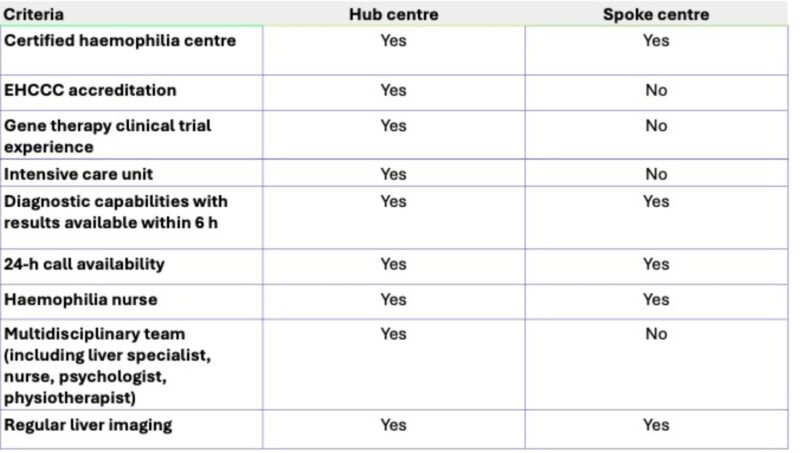
Hub-and-spoke model: The publication details a scalable and flexible framework—hub centres manage dosing and complexity while spoke centres provide local follow-up—making advanced therapies feasible and accessible across diverse care settings.
Unprecedented multidisciplinary team requirements including gene therapy liaison nurses, specialized pharmacists, administrative coordinators, and mandatory access to hepatologists.
Clear operational standards: The authors provide actionable criteria (e.g., bioisolator for ATMPs, 24-hour on-call service, rapid laboratory turnaround) and delineate staff roles, allowing centres to objectively assess readiness.
Flexible and adaptive: The model accommodates regional differences and supports spoke centre capacity-building, allowing responsibility to shift as local experience increases—a practical approach for evolving health systems.
Focus on safety and coordination: Emphasizes standardized protocols, scenario preparedness (“dry run” exercises), and robust SOPs for adverse event management, underpinning a safety-first transition to gene therapy.
Globally relevant: While based on European experience, the findings and framework are adaptable to international settings, with comparisons to other models such as the US health system.
Congratulations and a BIG THANK YOU to the EAHAD Gene Therapy Working Group!”
For more information follow the link.
Stay informed with Hemostasis Today.
-
May 7, 2026, 17:56The Latest Clinical Data Shaping The Evolving Landscape of Hemophilia Treatment – EAHAD
-
May 7, 2026, 17:31Talha Badar: New Classical Hematology Chapter on Bleeding and Thrombosis
-
May 7, 2026, 17:26Maria Τektonidou: Gene Network Analysis Identifies STAT1 in Thrombotic APS
-
May 7, 2026, 17:23Sonu Bhaskar: Inflammatory Biomarkers in Acute Cerebral Venous Sinus Thrombosis
-
May 7, 2026, 16:53Patricio S. Espinosa: Anticoagulation Is Not an Absolute Contraindication to Amyloid Therapy
-
May 7, 2026, 16:34Arun V J: Give Blood and Be the Hero You Always Wanted to Be
-
May 7, 2026, 16:15August Bones: When Would You Consider Placing an Inferior Vena Cava Filter After a Pulmonary Embolism?
-
May 7, 2026, 15:58Stop Asking Doctors to Do More – Reimagining the Care Team – The US Oncology
-
May 7, 2026, 15:42Chokri Ben Lamine: CVC-Associated UE-DVT and Catheter Removal

