
Kanishk Kumar: ATTR Amyloidosis Therapeutics Market Poised for a Pipeline Revolution
Kanishk Kumar, Digital Marketing Executive at DelveInsight Business Research LLP, shared a post on LinkedIn:
”The ATTR amyloidosis treatment landscape is undergoing rapid transformation, led by innovators such as Alnylam Pharmaceuticals-US , Novo Nordisk , SIRNAOMICS , Arbor Biotechnologies. With the rise of RNA interference (RNAi) therapies, TTR stabilizers, gene-editing platforms, and next‑generation silencers, these companies are redefining standards of care. Development efforts are focused on slowing or halting disease progression, improving quality of life, and extending survival for both hereditary (hATTR) and wild-type (wtATTR) amyloidosis. As increasingly precise, targeted approaches advance, the pipeline is set to usher in a new era of personalized medicine for a condition once considered largely untreatable.
DelveInsight’s ‘ATTR Amyloidosis Pipeline Insight, 2026’ provides an in‑depth evaluation of the current clinical landscape and future growth potential in the ATTR amyloidosis market. The report includes disease background, current treatment guidelines, and a detailed review of the pipeline from preclinical through marketed stages. It covers mechanisms of action, clinical trial data, regulatory status, and key development activities such as collaborations, mergers and acquisitions, funding, and special designations.
For emerging ATTR amyloidosis therapies, the report delivers a 360° view of the competitive and scientific landscape, segmented by development stage, product type, route of administration, molecule type, and mechanism of action (MOA). It also examines business opportunities, challenges, potential partnerships, competitive intensity, and strategic growth pathways.
Key Takeaways from the ATTR Amyloidosis Pipeline Report
- DelveInsight’s analysis shows a robust field, with 10+ active companies developing 10+ pipeline candidates for ATTR amyloidosis.
- Leading players include Alnylam Pharmaceuticals-US , Novo Nordisk , SIRNAOMICS , Arbor Biotechnologies , and others working to advance next‑generation therapies.
- Notable pipeline assets in various stages of development include Vutrisiran, NN‑6019, ALN‑TTRsc04, STP152G, ABO‑102, among others.
- In March: Alnylam Pharmaceuticals presented moderated posters featuring new data from the Phase III HELIOS‑B trial of vutrisiran for ATTR amyloidosis with cardiomyopathy (ATTR‑CM), in a patient cohort representative of real‑world practice.
- In November :Alnylam announced FDA acceptance of its supplemental new drug application (sNDA) for Amvuttra (vutrisiran), seeking label expansion to include ATTR‑CM.
Request a sample and explore the latest advances in the ATTR amyloidosis pipeline.
ATTR Amyloidosis Overview
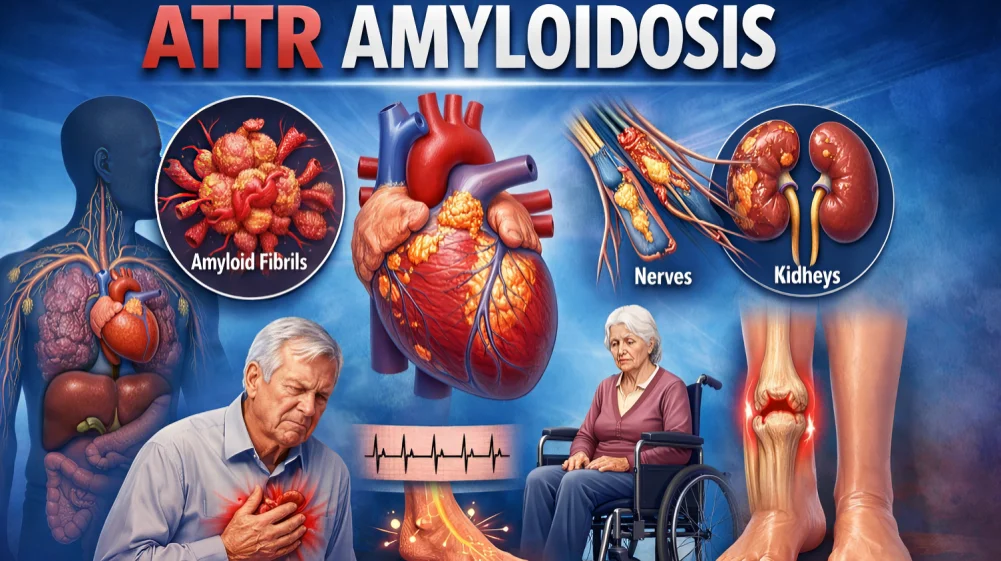
Transthyretin amyloidosis (ATTR)—often referred to in its cardiac form as ATTR cardiomyopathy (ATTR‑CM)—is characterized by the misfolding of the transthyretin (TTR) protein and its deposition as amyloid fibrils in tissues, most notably the heart. TTR, primarily synthesized by the liver, normally transports retinol (vitamin A) and thyroxine (T4). Structural alterations in TTR destabilize the tetramer, leading to misfolding, aggregation, and amyloid deposition.
These deposits commonly affect the myocardium and peripheral nerves, giving rise to symptoms such as progressive heart failure, neuropathic pain, weakness, and impaired temperature or sensory perception. In hereditary ATTR (ATTR), symptoms typically appear between the mid‑20s and mid‑60s and worsen over time. Diagnosis usually requires tissue biopsy with histopathology and immunohistochemistry, though extracardiac biopsy samples may have variable sensitivity.
Learn more about ATTR amyloidosis treatments.
ATTR Amyloidosis Treatment Analysis: Key Drug Profiles
Vutrisiran – Alnylam Pharmaceuticals
Vutrisiran is an investigational subcutaneously administered RNAi therapeutic designed to treat both hereditary and wild‑type ATTR amyloidosis. It selectively targets TTR messenger RNA, suppressing hepatic production of TTR protein and thereby aiming to prevent further amyloid fibril formation. With dosing every three or six months, vutrisiran is intended to reduce amyloid burden and help improve or preserve organ function.
The therapy leverages Alnylam’s Enhanced Stabilization Chemistry (ESC) GalNAc-conjugate platform, which enhances potency and metabolic stability, enabling infrequent subcutaneous dosing. Vutrisiran remains under investigation and has not yet been fully evaluated by regulatory authorities for all indications; it is currently in Phase III development for ATTR amyloidosis with cardiomyopathy.
NN‑6019 – Novo Nordisk
NN‑6019 is an investigational humanized monoclonal antibody developed to selectively recognize and clear amyloid deposits in both hereditary and wild‑type ATTR (hATTR and wtATTR), without disrupting the normal, functional tetrameric form of TTR. Preclinical research has shown that NN‑6019 facilitates removal of insoluble amyloid fibrils via antibody-mediated phagocytosis and also inhibits further amyloid formation.
This mechanism offers the potential to benefit patients at high risk of early mortality due to extensive amyloid deposition in critical organs. NN‑6019 is currently in Phase II clinical evaluation for ATTR amyloidosis.
Discover more about emerging ATTR amyloidosis therapies.
ATTR Amyloidosis Therapeutic Assessment
By Product Type
- Monotherapy
- Combination therapy
- Mono/Combination
By Development Stage
- Late‑stage products (Phase III)
- Mid‑stage products (Phase II)
- Early‑stage products (Phase I)
- Preclinical and Discovery-stage candidates
- Discontinued & Inactive programs
By Route of Administration
- Oral
- Intravenous
- Subcutaneous
- Parenteral
- Topical
By Molecule Type
- Recombinant fusion proteins
- Small molecules
- Monoclonal antibodies
- Peptides
- Polymers
- Gene therapies
Scope of the ATTR Amyloidosis Pipeline Report
- Coverage: Global
- Key Companies: Alnylam Pharmaceuticals-US , Novo Nordisk , SIRNAOMICS , Arbor Biotechnologies, and others.
- Key Pipeline Therapies: Vutrisiran, NN‑6019, ALN‑TTRsc04, STP152G, ABO‑102, and additional candidates.
For detailed, product‑level insights on ATTR amyloidosis drugs.
Table of Contents (Report Structure)
- Introduction
- Executive Summary
- ATTR Amyloidosis Pipeline: Overview
- Analytical Perspective – In‑depth Commercial Assessment
- ATTR Amyloidosis Pipeline Therapeutics
- ATTR Amyloidosis Pipeline: Late‑Stage Products (Phase III)
- ATTR Amyloidosis Pipeline: Mid‑Stage Products (Phase II)
- ATTR Amyloidosis Pipeline: Early‑Stage Products (Phase I)
- Therapeutic Assessment
- Inactive Products
- Company–University Collaborations (Licensing/Partnering) Analysis
- Key Companies
- Key Products
- Unmet Needs
- Market Drivers and Barriers
- Future Perspectives and Conclusion
- Analyst Views
- Appendix
About DelveInsight
DelveInsight is a premier healthcare-oriented market research and consulting firm dedicated to delivering high-caliber market intelligence and analytical insights that empower informed business decision-making. Supported by a team of seasoned industry specialists with deep expertise across the life sciences and healthcare domains, DelveInsight provides tailored research solutions and strategic guidance to clients worldwide. Partner with us to access high-quality, precise, and real-time intelligence that keeps you ahead of the competitive curve.”
Stay updated with Hemostasis Today.
-
May 12, 2026, 16:46Tagreed Alkaltham: Why Apheresis Matters in Modern Transfusion Medicine
-
May 12, 2026, 16:37Reinhold Kreutz: Cardiovascular Burden in Acute Intermittent Porphyria Needs Greater Awareness
-
May 12, 2026, 16:33Pablo Corral: The Truth About Very Low LDL-Cholesterol
-
May 12, 2026, 16:24Mildred Lundgren: We Must Talk About the Invisible Causes of Stroke
-
May 12, 2026, 16:17Irene Scala: The Sex Disparities In Access to Acute Stroke Treatments In Italy
-
May 12, 2026, 16:04May Nour: UCLA Health Mobile Stroke Unit Becomes The 1st In The World to Perform mCTA In the Field
-
May 12, 2026, 15:57Leonardo Roever: Prognostic Impact of Lipoprotein(a) and CAR in Elderly Acute Ischemic Stroke Patients
-
May 12, 2026, 15:54Bruno Pougault: Prioritizing Laboratory Tests in Resource-Limited Emergency Care
-
May 12, 2026, 15:37Jennifer Holter Chakrabarty: Supporting the Next Generation of Hematology Researchers

