
Dan Purser: MTHFR Mutations and Elevated Homocysteine in Autoimmune Inflammation
Dan Purser, Co-Founder of AO Biologix, LLC, shared a post on LinkedIn:
“For more than four decades as a practicing physician, I have watched thousands of patients cycle through conventional autoimmune treatments that address symptoms but never touch the fire underneath.
Rheumatoid arthritis flares, lupus rashes, multiple sclerosis relapses, Hashimoto’s thyroiditis, fibromyalgia pain—these are not random.
They share a common, often overlooked biochemical pathway:
MTHFR mutations leading to elevated homocysteine that supercharges autoimmune inflammation.
This is not fringe theory.
It is the clinical reality I have mapped in my practice, refined over 12 years of data from thousands of patients, and will soon publish in my 600-page textbook Homocysteine: The 2026 Comprehensive Diagnosis and Treatment Protocol.
My work is a decade ahead of mainstream medicine because I treat the root—not the flare.
And today I am sharing the complete Purser Method framework so you (or your patients) can finally break the cycle.
The Silent Epidemic:
MTHFR, Homocysteine, and the Autoimmune Storm
Approximately 85% of the U.S. population carries at least one MTHFR polymorphism (most commonly C677T or A1298C).
These single-nucleotide changes reduce the enzyme methylenetetrahydrofolate reductase activity by 30–70%, impairing the conversion of folate into its active form (5-methyltetrahydrofolate).
The downstream result?
Impaired methylation, elevated plasma homocysteine, and a body-wide amplification of inflammatory cytokines.
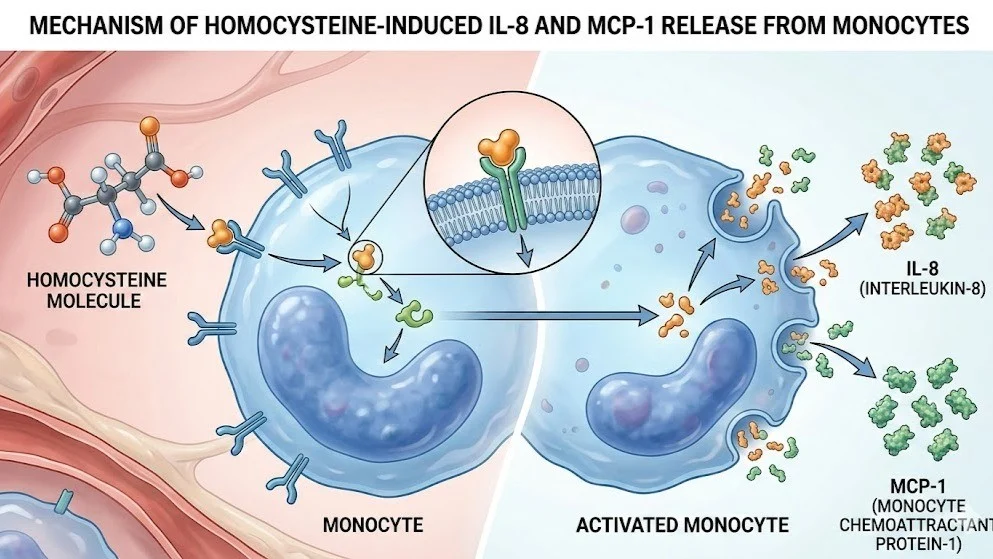
Homocysteine is not merely a cardiovascular marker.
It is a potent ‘cytokine amplifier.’
When levels climb above 10–12 µmol/L (my functional optimum is lower7–8 µmol/L), homocysteine directly stimulates production of IL-6, TNF-α, IL-17, IL-1β, IL-8, and MCP-1—the exact Th17/Th1-dominant storm driving most autoimmune diseases.
Recent meta-analyses confirm the link:
- MTHFR C677T and A1298C polymorphisms increase genetic susceptibility to rheumatoid arthritis (especially in Asian populations) and systemic lupus erythematosus.
- In multiple sclerosis, the MTHFR 677TT genotype correlates with higher homocysteine, greater disability progression, and elevated Th17 cytokines (IL-17, IL-6, TNF-α).
- Hyperhomocysteinemia is documented in ankylosing spondylitis, juvenile idiopathic arthritis, and Hashimoto’s, with homocysteine levels directly tracking disease activity.
I have seen this in my clinic for years.
- A 42-year-old woman with ‘seronegative RA’ and sky-high IL-6 despite biologics.
- A 35-year-old with relapsing optic neuritis and NMOSD-like cytokine profile.
- A 28-year-old with refractory Hashimoto’s and brain fog.
All had compound heterozygous MTHFR and homocysteine higher than 15 µmol/L.
When we corrected the methylation block, their cytokine storms quieted and symptoms melted away.
This is why I call homocysteine the ‘hidden root cause most doctors miss.’
Conventional rheumatologists measure ESR, CRP, and autoantibodies—but rarely homocysteine or MTHFR.
They prescribe immunosuppressants that further deplete folate and raise homocysteine, creating a vicious cycle.
My Purser Method breaks it.
The Science:
How MTHFR-Driven Homocysteine Ignites Autoimmune InflammationLet me walk you through the exact biochemistry – because understanding it empowers healing.
- MTHFR Block – Homocysteine Buildup The C677T variant (especially homozygous TT) reduces enzyme activity up to 70%. A1298C compounds the effect. Result: homocysteine cannot be efficiently remethylated to methionine or transsulfurated to cysteine/glutathione.
- Homocysteine as Pro-Inflammatory Trigger Elevated homocysteine activates NF-κB, upregulates adhesion molecules, and directly induces IL-6, TNF-α, IL-17, and IL-1β in macrophages and T-cells. It also promotes oxidative stress and endothelial dysfunction, allowing immune cells to infiltrate tissues.
- Th17 Skewing and Autoantibody Production Homocysteine drives naive T-cells toward Th17 differentiation via IL-6 and IL-23 signaling. Th17 cells release IL-17, recruiting neutrophils and amplifying tissue destruction—classic in RA synovitis, MS plaques, lupus nephritis, and even MOGAD optic neuritis.
- Glutathione Collapse The transsulfuration pathway is blocked, so glutathione (your master antioxidant) plummets. Without glutathione, oxidative stress skyrockets, further fueling cytokine production and autoimmunity.
This pathway explains why standard autoimmune therapies often fail long-term.
They suppress downstream cytokines but never fix the upstream methylation defect.
My patients on the Purser Method achieve remission rates that conventional protocols cannot match—because we treat the genetic root.
Visual Summary of Cytokine Profiles in Optic Neuritis (MS vs NMOSD vs MOGAD)[Embed the triptych cytokine diagram here].”
Stay updated with Hemostasis Today.
-
May 12, 2026, 16:46Tagreed Alkaltham: Why Apheresis Matters in Modern Transfusion Medicine
-
May 12, 2026, 16:37Reinhold Kreutz: Cardiovascular Burden in Acute Intermittent Porphyria Needs Greater Awareness
-
May 12, 2026, 16:33Pablo Corral: The Truth About Very Low LDL-Cholesterol
-
May 12, 2026, 16:24Mildred Lundgren: We Must Talk About the Invisible Causes of Stroke
-
May 12, 2026, 16:17Irene Scala: The Sex Disparities In Access to Acute Stroke Treatments In Italy
-
May 12, 2026, 16:04May Nour: UCLA Health Mobile Stroke Unit Becomes The 1st In The World to Perform mCTA In the Field
-
May 12, 2026, 15:57Leonardo Roever: Prognostic Impact of Lipoprotein(a) and CAR in Elderly Acute Ischemic Stroke Patients
-
May 12, 2026, 15:54Bruno Pougault: Prioritizing Laboratory Tests in Resource-Limited Emergency Care
-
May 12, 2026, 15:37Jennifer Holter Chakrabarty: Supporting the Next Generation of Hematology Researchers

