
Abdulla A. Damluji: Pulsed Field Ablation Delivers a New Standard for Persistent AF
Abdulla A. Damluji, Director of the Cardiovascular Center on Aging at Cleveland Clinic, shared on LinkedIn about a recent article by Oussama M. Wazni et al, published in NEJM, adding:
“Pulsed Field Ablation as First Line Therapy for Persistent Atrial Fibrillation: The AVANT GUARD Trial
Atrial fibrillation affects an estimated 52.5 million people worldwide and is associated with thromboembolism, cognitive dysfunction, heart failure, premature death, and substantial use of arrhythmia related health care.
Persistent atrial fibrillation, defined as a continuous episode lasting more than 7 days, accounts for up to 50% of patients with the condition and carries a higher burden of adverse outcomes than paroxysmal disease.
Although catheter ablation is established as first line therapy for paroxysmal atrial fibrillation, current guidelines still recommend a trial of antiarrhythmic drug therapy before ablation in persistent disease, in part because thermal ablation has shown inferior outcomes in atria with greater electrical and structural remodeling.
The AVANT GUARD trial, published in the New England Journal of Medicine on April 25, 2026, was an international, multicenter, randomized controlled trial sponsored by Boston Scientific.
The trial evaluated whether initial rhythm control with pulsed field ablation using the pentaspline FARAWAVE catheter is superior to guideline directed antiarrhythmic drug therapy in patients with previously untreated persistent atrial fibrillation.
Adults with symptomatic, persistent atrial fibrillation lasting 7 to 365 days were eligible. Patients with prior exposure to class one or class three antiarrhythmic drugs for more than 7 consecutive days, or to amiodarone for more than 24 hours within 6 months, were excluded.
A total of 310 patients underwent randomization in a 2 to 1 ratio, with 207 assigned to pulsed field ablation and 103 to antiarrhythmic drug therapy. An additional 100 patients were assigned to pulsed field ablation for the primary safety analysis.
Patients in the ablation group underwent pulmonary vein isolation and posterior wall ablation with confirmation of electrical isolation after a 20 minute waiting period.
Patients in the drug group received guideline directed therapy, with flecainide prescribed in 57.2% at a median dose of 200 milligrams per day and sotalol in 28.2% at a median dose of 240 milligrams per day.
Cardioversion was performed in 64.2% of patients in the drug group. All patients received an insertable cardiac monitor (LUX-Dx, Boston Scientific) with continuous arrhythmia detection by the BeatLogic algorithm.
The trial was temporarily paused on October 18, 2024 after 6 neurologic events.
Enrollment resumed on November 7, 2024 with protocol modifications, including exclusion of patients with a CHA2DS2-VASc score of 4 or higher, mandatory pre-procedural screening for left atrial thrombus by transesophageal echocardiography or computed tomography, uninterrupted anticoagulation for at least 4 weeks before the procedure, and a minimum activated clotting time of 350 seconds before ablation.
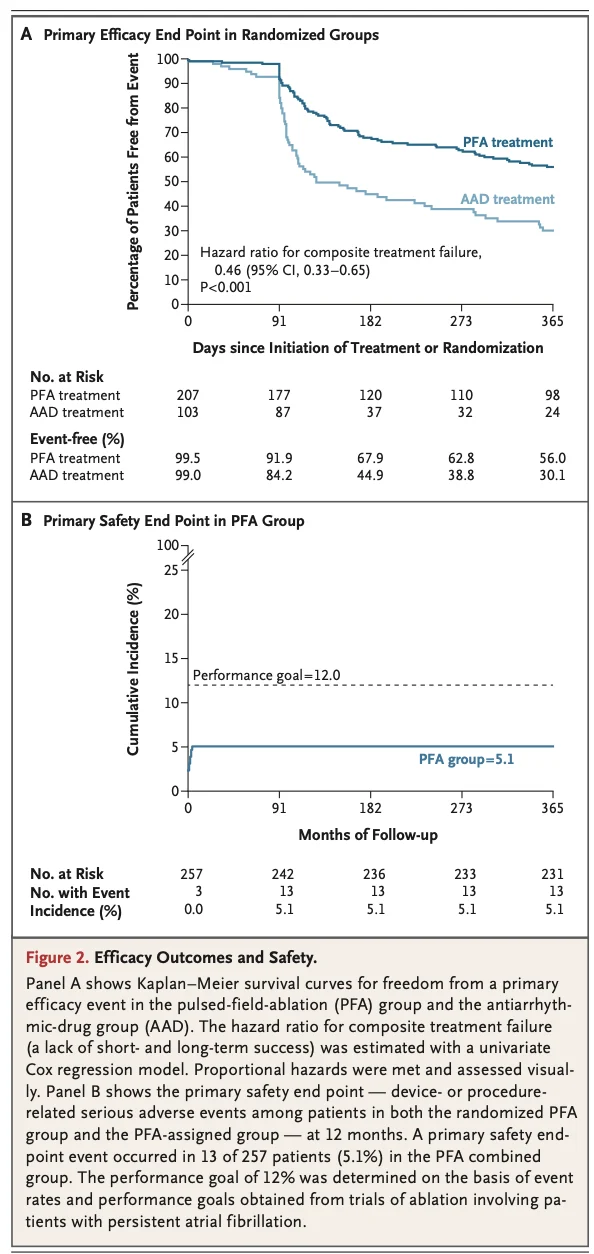
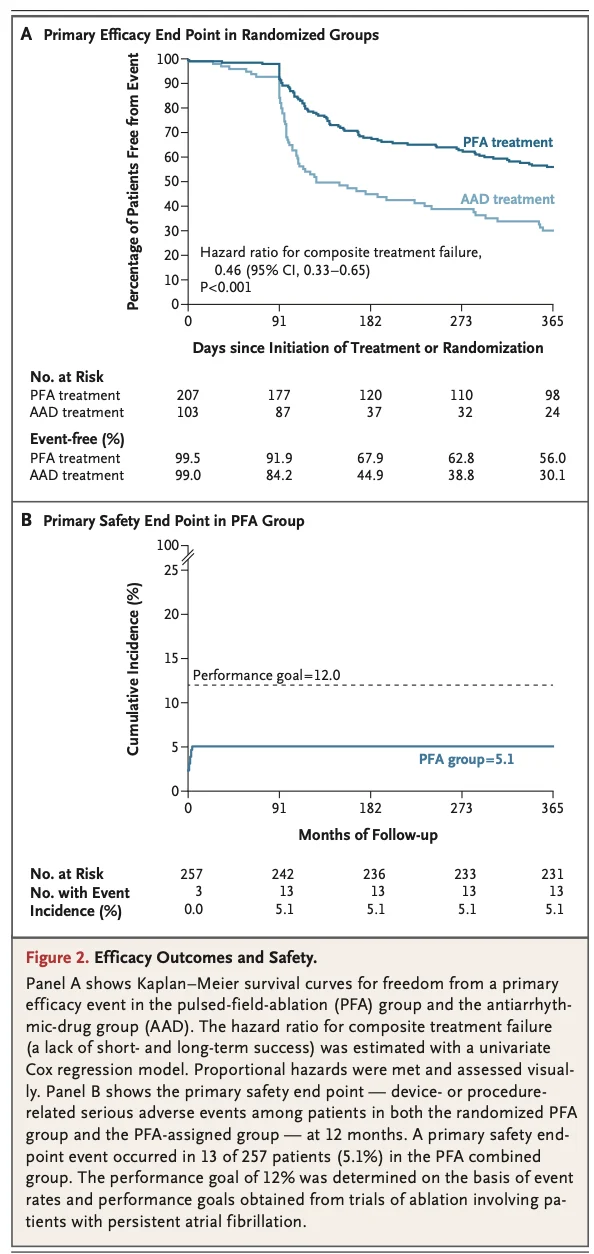
At 12 months, the primary effectiveness endpoint of short and long term success occurred in 128 of 207 patients in the pulsed field ablation group (Kaplan-Meier estimate 56%; 95% confidence interval 48 to 63) compared with 40 of 103 patients in the antiarrhythmic drug group (Kaplan-Meier estimate 30%; 95% confidence interval 21 to 40).
The absolute difference was 26 percentage points (95% confidence interval 14 to 38), and the hazard ratio for composite treatment failure was 0.46 (95% confidence interval 0.33 to 0.65; P less than 0.001).
The treatment effect appeared consistent across prespecified subgroups, as shown in Table 2 and Figure 2A.
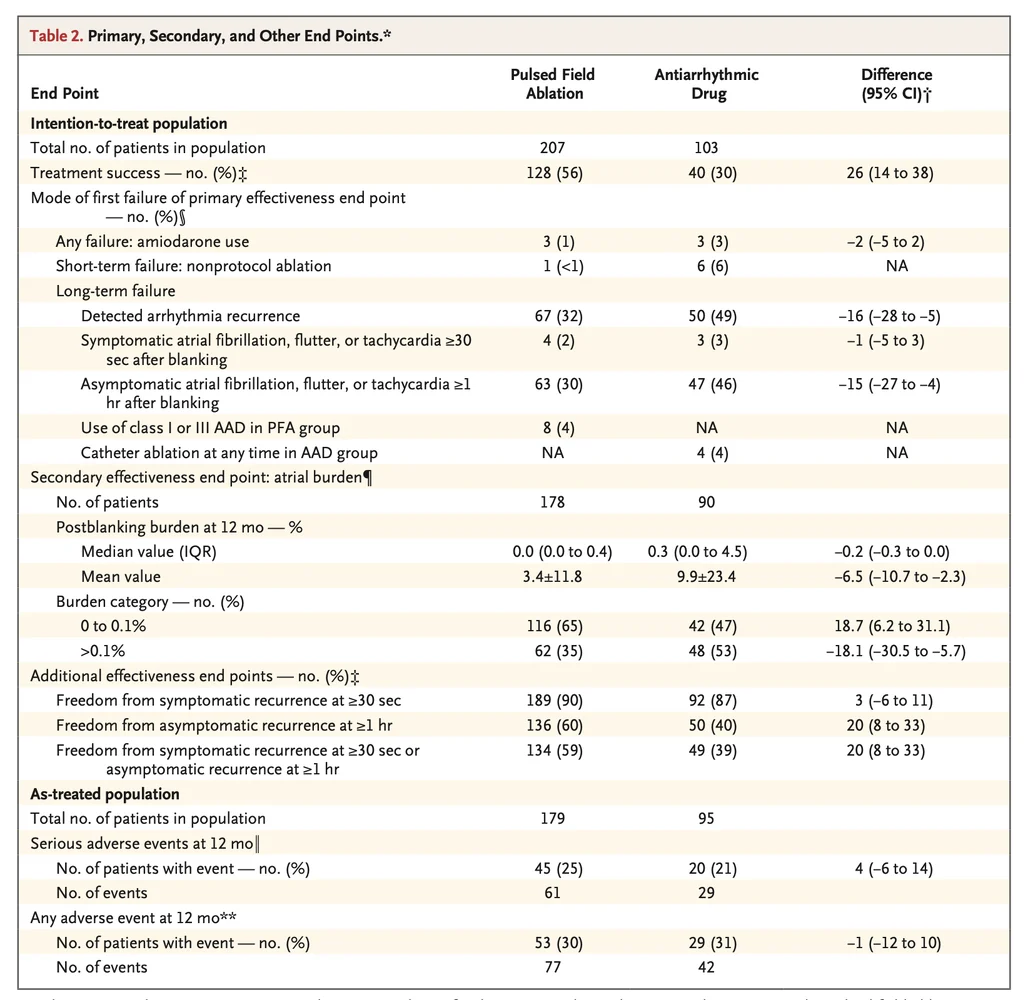
Recurrence of symptomatic atrial tachyarrhythmia of at least 30 seconds occurred in 2% of ablation patients and 3% of drug therapy patients, and asymptomatic recurrence of at least 1 hour occurred in 30% versus 46%, respectively.
Median atrial arrhythmia burden at 1 year was 0% in the ablation group and 0.2% in the drug group. Nonprotocol ablation was performed in less than 1% of ablation patients and 6% of drug therapy patients.
Quality of life as measured by the Atrial Fibrillation Effect on Quality-of-Life survey improved in both groups, with mean changes from baseline of 23.3 in the ablation group and 19.8 in the drug therapy group at 12 months.
The Kaplan-Meier incidence of the primary safety endpoint, defined as device or procedure related serious adverse events, was 5.1% with an upper confidence limit of 8.6%, meeting the prespecified performance goal of less than 12% (Table 3, Figure 2B).
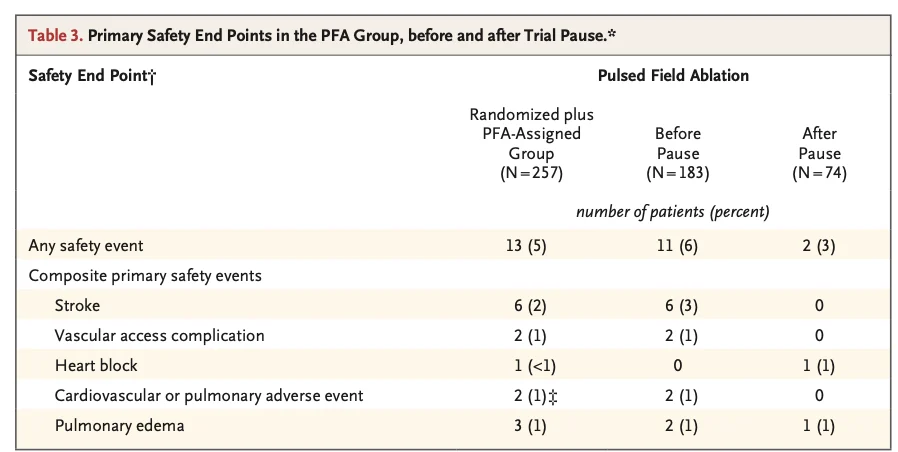
Six procedure related strokes were adjudicated, one related to inadvertent introduction of air from the deflectable sheath. All strokes were graded as National Institutes of Health Stroke Scale less than 5 with subsequent clinical improvement or full recovery.
Safety event rates in the ablation group were 6% before and 3% after the protocol modification, with no strokes or transient ischemic attacks observed after the pause. At 12 months, serious adverse events of any cause occurred in 25% of the ablation group and 21% of the drug therapy group.
The trial has several limitations. The follow up was 12 months, and longer term outcomes will require additional study. The trial used a single ablation technique with a standard lesion set, so the findings may not generalize to other catheters, energy sources, or strategies.
The cardiac monitor was inserted at the time of treatment initiation, precluding assessment of change in arrhythmia burden from baseline. Some patients in the drug group underwent ablation after pharmacologic failure, which may have led to underestimation of the effect of ablation on arrhythmia burden.
The primary safety endpoint was evaluated against a performance goal rather than directly compared with the drug therapy group, given the inherent differences in adverse event timing between procedures and pharmacotherapy.
The trial population after the pause represented only 29% of treated patients, limiting attribution of the post pause safety improvement to procedural versus patient factors.
In patients with previously untreated persistent atrial fibrillation, first line treatment with pulsed field ablation using the pentaspline catheter was superior to guideline directed antiarrhythmic drug therapy for the maintenance of sinus rhythm at 12 months, with a similar incidence of serious adverse events between groups.
The findings support consideration of pulsed field ablation as an initial treatment strategy in appropriately selected patients, while reinforcing the importance of careful periprocedural management in older patients with multiple coexisting illnesses.
Key Points Helpful for Clinical Practice
- In patients with previously untreated persistent atrial fibrillation, first line pulsed field ablation with the pentaspline catheter resulted in a 12 month treatment success rate of 56% compared with 30% for antiarrhythmic drug therapy (hazard ratio for treatment failure 0.46; P less than 0.001).
- The atrial arrhythmia burden as measured by an insertable cardiac monitor was significantly lower in the ablation group, with median values of 0% versus 0.2% and a mean between group difference of 6.5 percentage points.
- Serious adverse events of any cause at 12 months were similar between groups (25% ablation versus 21% drug therapy), and the device or procedure related serious adverse event rate of 5.1% met the prespecified safety performance goal.
- Six procedure related strokes were observed, prompting a temporary trial pause and protocol modifications including stricter anticoagulation, pre-procedural imaging, and exclusion of patients with CHA2DS2-VASc score of 4 or higher; no strokes occurred after these modifications.
- The findings apply to patients with persistent atrial fibrillation lasting 7 to 365 days who have not received prolonged class one, class three, or amiodarone therapy; clinicians should weigh the periprocedural risks against the rhythm control benefit through shared decision making, particularly in older patients with multiple coexisting illnesses.
Key Market Implications
- AVANT GUARD provides Boston Scientific with the first randomized controlled evidence supporting FARAPULSE as a first line option in persistent atrial fibrillation, an indication that has historically required documented antiarrhythmic drug failure before ablation. This positions the FARAWAVE pentaspline catheter ahead of competing pulsed field ablation platforms, including the Medtronic PulseSelect and Affera Sphere-9, the Johnson and Johnson Varipulse, and the Abbott Volt systems, none of which currently have published randomized first line persistent atrial fibrillation data of comparable scope.
- The trial materially expands the addressable patient population for catheter ablation. Persistent atrial fibrillation accounts for up to 50% of the estimated 52.5 million patients with atrial fibrillation worldwide, and a guideline shift toward first line ablation would move large numbers of patients out of the antiarrhythmic drug treatment phase into the procedural pathway, increasing case volume for electrophysiology programs and procedural revenue for device manufacturers.
- Updated practice guidelines from the American College of Cardiology, American Heart Association, Heart Rhythm Society, and European Society of Cardiology are likely to incorporate these data in upcoming revisions, with a probable upgrade in the recommendation class for first line ablation in persistent atrial fibrillation. Such a change would have downstream effects on payer coverage policies and prior authorization requirements.
- The antiarrhythmic drug market for atrial fibrillation, including flecainide, sotalol, dronedarone, and propafenone, may experience reduced first line utilization in persistent disease, with corresponding pressure on revenue from manufacturers of these agents. The 6% rate of nonprotocol ablation in the drug therapy group within 12 months also points to limited durability of pharmacologic rhythm control as a long term strategy.
- The integrated use of the LUX-Dx insertable cardiac monitor with the BeatLogic algorithm strengthens Boston Scientific’s combined ablation and monitoring offering and sets a higher bar for outcome measurement in future ablation trials. Continuous monitoring with quantitative arrhythmia burden may become an expected standard for trial design and post procedural surveillance, benefiting incumbent insertable cardiac monitor manufacturers including Medtronic (Reveal LINQ) and Abbott (Confirm Rx).
- The 5.1% device or procedure related serious adverse event rate, including the cluster of neurologic events that prompted the trial pause, will sharpen scrutiny of periprocedural anticoagulation, imaging, and activated clotting time protocols across the field. Manufacturers and electrophysiology programs that adopt the post pause workflow proactively are positioned to defend safety claims as pulsed field ablation expands into older and higher risk populations.
- The trial’s older and more comorbid population than prior ablation cohorts supports broader real world generalizability and may accelerate adoption in community electrophysiology practices, increasing demand for operator training, capital equipment placements, and disposable catheter sales over the next 24 to 36 months.”
Title: Pulsed Field Ablation as Initial Therapy for Persistent Atrial Fibrillation
Authors: Oussama M. Wazni, K.R. Julian Chun, Devi G. Nair, Ante Anic, Mattias Duytschaever, Jonathan Chrispin, Anish Amin, Olujimi A. Ajijola, Douglas N. Gibson, Moussa Mansour, Jonathan P. Piccini, Andrea Natale, Auroa Badin, Joaquin Osca, Jonathan Dukes, Karan Bhalla, Ayman Hussein, Todd Senn, Stanislav Weiner, Nitesh Sood, Antonio Dello Russo, Ashish Sadhu, Jason Meyers, Kevin Trulock, Mark T.K. Tam, David B. DeLurgio, Jean Champagne, Saverio Iacopino, Brian Kwan, Elizabeth M. Albrecht, Sheryl McCammon, Ann C. Garlitski, Brad S. Sutton, Kenneth M. Stein, Jason G. Andrade

Stay updated on all scientific advances with Hemostasis Today.
-
May 12, 2026, 16:46Tagreed Alkaltham: Why Apheresis Matters in Modern Transfusion Medicine
-
May 12, 2026, 16:37Reinhold Kreutz: Cardiovascular Burden in Acute Intermittent Porphyria Needs Greater Awareness
-
May 12, 2026, 16:33Pablo Corral: The Truth About Very Low LDL-Cholesterol
-
May 12, 2026, 16:24Mildred Lundgren: We Must Talk About the Invisible Causes of Stroke
-
May 12, 2026, 16:17Irene Scala: The Sex Disparities In Access to Acute Stroke Treatments In Italy
-
May 12, 2026, 16:04May Nour: UCLA Health Mobile Stroke Unit Becomes The 1st In The World to Perform mCTA In the Field
-
May 12, 2026, 15:57Leonardo Roever: Prognostic Impact of Lipoprotein(a) and CAR in Elderly Acute Ischemic Stroke Patients
-
May 12, 2026, 15:54Bruno Pougault: Prioritizing Laboratory Tests in Resource-Limited Emergency Care
-
May 12, 2026, 15:37Jennifer Holter Chakrabarty: Supporting the Next Generation of Hematology Researchers

