
Amir Kraitzer: Engineering Hemocompatible Surfaces to Overcome Thrombosis in Medical Devices
Amir Kraitzer, Independent MedTech and Innovation Consultant, shared a post on LinkedIn:
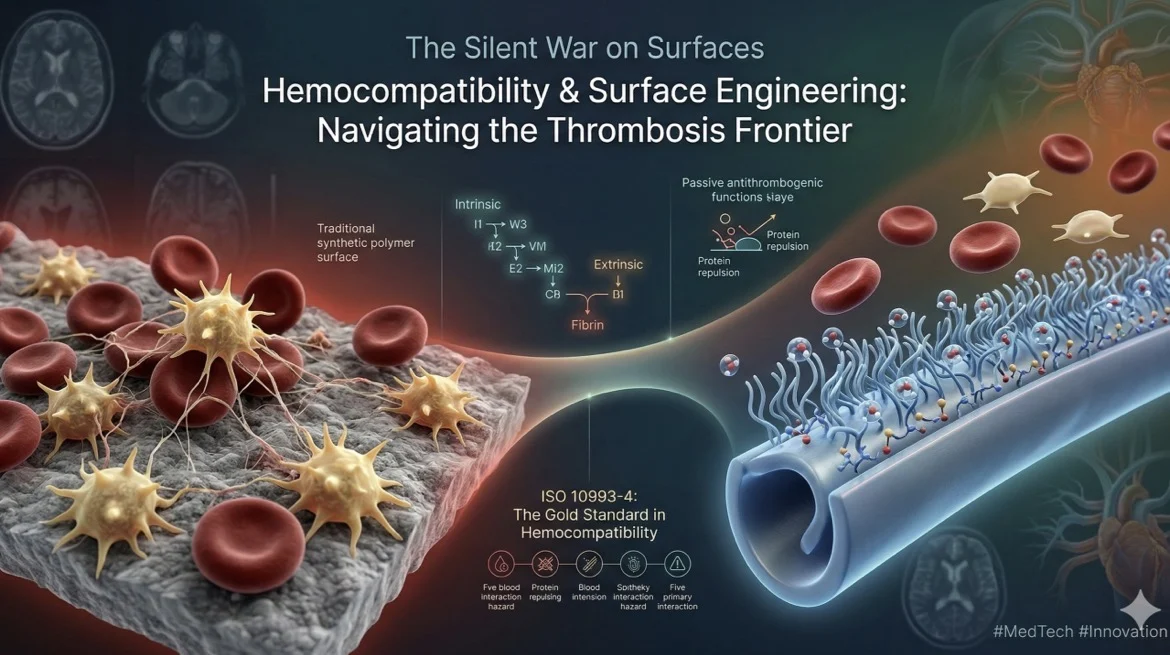
“Each year, millions of clinical procedures require the use of blood-contacting medical devices, including stents, catheters, and extracorporeal circuits. Despite significant engineering advancements, these devices remain subject to a fundamental physiological challenge: thrombosis. The intersection of material science and hematology is where the next generation of patient outcomes will be decided.
The Bio-Interface: Mechanisms of Coagulation
The human vascular system is lined with a quiescent monolayer of endothelial cells that actively inhibits thrombus formation through the production of anticoagulant factors. Synthetic materials lack these active inhibitory functions, triggering a complex hemostatic response upon contact:
- Platelet Adhesion and Activation: Foreign surfaces provide a scaffold for platelet attachment, initiating structural changes and the release of procoagulant granules.
- The Coagulation Cascade: Activation of the intrinsic pathway engages a series of coagulation factors, culminating in the conversion of prothrombin to enzymatically active thrombin.
- Fibrin Polymerization: Thrombin transforms soluble fibrinogen into an insoluble fibrin mesh, which, in conjunction with activated platelets, stabilizes the thrombus.
- Complement Activation: The immune system identifies artificial surfaces as foreign, triggering a complement-mediated inflammatory response that can complicate clinical recovery.
Regulatory Compliance
To ensure patient safety, all antithrombogenic coatings must undergo rigorous evaluation according to ISO 10993-4. This standard provides a structured framework for identifying interaction hazards across five primary categories:
- Thrombosis (Clot formation)
- Hemolysis
- Platelet Activation and Adhesion
- Complement System Activation
- Coagulation Cascade Kinetics
This risk-based testing strategy is essential for the regulatory submission and clinical validation of cardiovascular grafts, heart valves, and dialysis equipment.
Current Strategies in Surface Modification
Modern antithrombogenic strategies aim to render device surfaces inert or bio-active to mitigate the blood-surface interface reaction:
- Heparin Immobilization: Technologies that utilize covalently bonded heparin to catalyze anti-thrombin activity, mimicking the natural anticoagulant properties of the vascular wall.
- Hydrophilic Polymer: These coatings generate a water-rich boundary layer, hydrogels, that physically repels protein adsorption, thereby reducing platelet adhesion.
- Biomimetic Phosphorylcholine (PC): PC-based polymers mimic the phospholipid headgroups of red blood cell membranes. Clinical data from PC coated devices demonstrate significantly reduced thrombin generation.
- Nitric Oxide (NO) release: NO release coatings replicate endothelial signaling to inhibit platelet activation, agrregation, and adhesion to the surface.
- Endothelial Progenitor cell (EPC) recruitment: Utilizes surface-bound antibodies (e.g., anti-CD34) to capture circulating EPCs. Once captured, these progenitor cells differentiate into a confluent, functional endothelial monolayer, establishing a living, auto-regenerative biological interface on the device surface.
The primary hurdle for long-term blood-contacting implants remains hemocompatibility. As medical technology advances, the efficacy of these devices will be defined by our success in engineering truly hemocompatible surfaces.”
Stay updated with Hemostasis Today.
-
May 12, 2026, 16:46Tagreed Alkaltham: Why Apheresis Matters in Modern Transfusion Medicine
-
May 12, 2026, 16:37Reinhold Kreutz: Cardiovascular Burden in Acute Intermittent Porphyria Needs Greater Awareness
-
May 12, 2026, 16:33Pablo Corral: The Truth About Very Low LDL-Cholesterol
-
May 12, 2026, 16:24Mildred Lundgren: We Must Talk About the Invisible Causes of Stroke
-
May 12, 2026, 16:17Irene Scala: The Sex Disparities In Access to Acute Stroke Treatments In Italy
-
May 12, 2026, 16:04May Nour: UCLA Health Mobile Stroke Unit Becomes The 1st In The World to Perform mCTA In the Field
-
May 12, 2026, 15:57Leonardo Roever: Prognostic Impact of Lipoprotein(a) and CAR in Elderly Acute Ischemic Stroke Patients
-
May 12, 2026, 15:54Bruno Pougault: Prioritizing Laboratory Tests in Resource-Limited Emergency Care
-
May 12, 2026, 15:37Jennifer Holter Chakrabarty: Supporting the Next Generation of Hematology Researchers

