
Asif Mushtaq: Managing Cardiac Surgery with Cardiopulmonary Bypass in Hemophilia
Asif Mushtaq, Chief Perfusionist at Punjab Institute of Cardiology, shared a post on LinkedIn:
“Introduction
Hemophilia is a hereditary bleeding disorder caused by deficiency of clotting factor VIII (Hemophilia A) or factor IX (Hemophilia B), resulting in impaired blood coagulation and a heightened risk of bleeding. Performing cardiac surgery requiring cardiopulmonary bypass (CPB) in hemophilia patients is particularly challenging. CPB necessitates systemic anticoagulation using heparin to prevent thrombosis in the extracorporeal circuit. This anticoagulation must be balanced carefully against the patient’s bleeding tendency due to factor deficiency. This article reviews the perioperative management strategies—focusing on heparin dosing, activated clotting time (ACT) monitoring, temperature management, and clotting factor replacement—and summarizes evidence from authentic clinical studies and guidelines.
Hemophilia and Cardiac Surgery: Specific Risks
CPB exposes blood to non-endothelial surfaces, activating coagulation pathways that require anticoagulation, traditionally with unfractionated heparin. In hemophilia patients, the underlying deficiency in clotting factors compounds the risk of bleeding. Surgical sites, cannulation points, and the cardiotomy reservoir are vulnerable to hemorrhage. Furthermore, hypothermia used during CPB for organ protection can impair coagulation enzymes and platelet function, worsening bleeding risk. Balancing thrombosis prevention in the circuit and minimizing patient bleeding is critical.
Perioperative Management Strategies
1. Clotting Factor Replacement Therapy
Before surgery, patients receive clotting factor concentrates to raise plasma factor levels to ≥80–100%, aiming to normalize coagulation as much as possible (World Federation of Hemophilia [WFH], 2020). During surgery, dosing is adjusted based on serial factor assays, with levels maintained near 100%. Postoperatively, factor replacement continues to maintain levels around 50–80% for 7 to 14 days until wound healing is adequate (WFH, 2020; Palla et al., 2017).
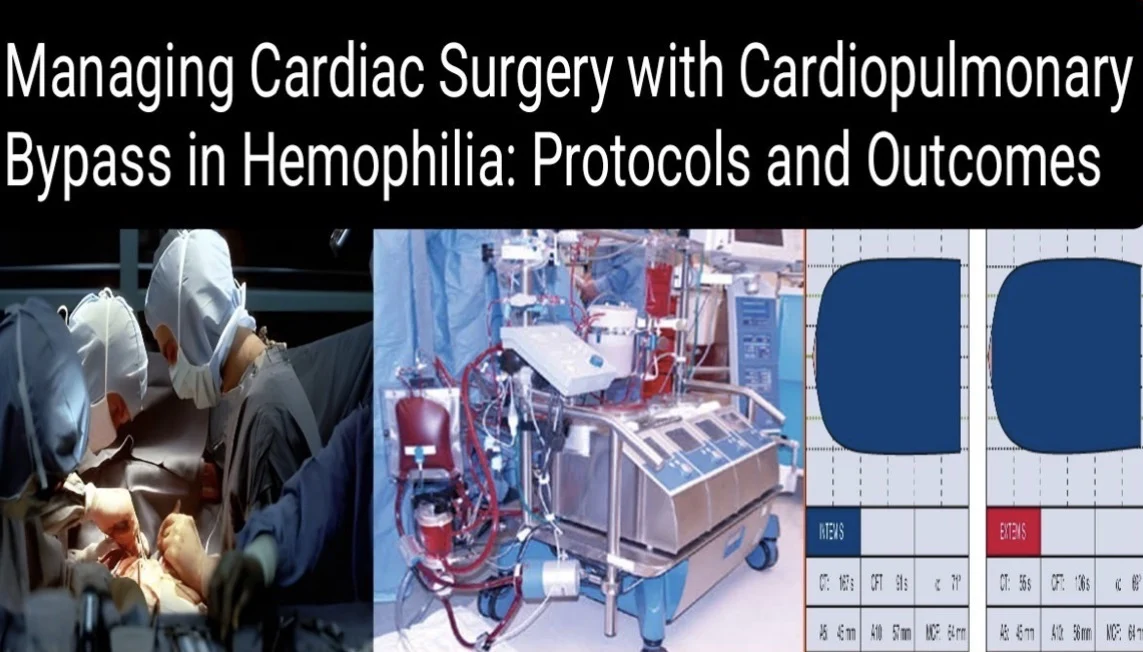
2. Heparin Dosing and ACT Monitoring
- Initial Heparin Dose: A bolus of 300–400 IU/kg intravenous unfractionated heparin is administered before aortic cannulation to anticoagulate the blood during CPB (Kobrinsky, 2016; Palla et al., 2017).
- ACT Monitoring: Baseline ACT is measured prior to heparin administration. After the bolus, ACT is checked 3–5 minutes later to ensure anticoagulation efficacy. The target ACT during CPB is >480 seconds to prevent clot formation in the circuit (Gandhi et al., 2019; WFH, 2020). ACT is measured every 30 minutes during CPB, with additional heparin doses (50–100 IU/kg) administered if ACT falls below target (Smith et al., 2019).
- Heparin Reversal: After CPB, protamine sulfate is given to neutralize heparin, typically at a dose of 1 mg protamine per 100 IU of heparin administered. ACT is monitored until it returns close to baseline (100–130 seconds) to confirm effective reversal (Kobrinsky, 2016; Palla et al., 2017).
3. Temperature Management
Temperature during CPB is carefully controlled. Mild hypothermia (28–34°C) is commonly used to reduce metabolic demand and protect organs. However, hypothermia impairs platelet function and coagulation enzyme activity, increasing bleeding risk (Shann et al., 2018). It also prolongs ACT independently of heparin effect, necessitating cautious interpretation of ACT values (Gandhi et al., 2019). Gradual rewarming to normothermia prior to weaning from CPB improves coagulation and reduces bleeding.
4. Adjunctive Therapies
Antifibrinolytic agents such as tranexamic acid are used to reduce fibrinolysis and help control bleeding.
5. Multidisciplinary Team Approach
Successful management requires close collaboration among hematologists, cardiac surgeons, anesthesiologists, perfusionists, and specialized nursing staff to monitor coagulation parameters, clinical bleeding, and factor levels continuously throughout the perioperative period.
Review of Clinical Evidence
- Palla et al. (2017): Reported a successful aortic valve replacement in a hemophilia A patient using intensive factor VIII replacement and heparin anticoagulation guided by ACT monitoring. Target ACT >480 seconds was maintained with protamine neutralization post-CPB, and no significant bleeding complications occurred.
- Smith et al. (2019): In a retrospective cohort of 12 hemophilia patients undergoing CPB cardiac surgery, meticulous heparin dosing, ACT monitoring every 30 minutes, and protamine reversal minimized bleeding complications with zero mortality.
- Gandhi et al. (2019): Demonstrated the effect of hypothermia on prolonging ACT independent of heparin, emphasizing the need for temperature-adjusted interpretation of ACT values during CPB.
- World Federation of Hemophilia (2020): Guidelines recommend maintaining clotting factor levels ≥80% perioperatively, heparin dosing protocols targeting ACT >480 seconds, and temperature control to optimize coagulation during cardiac surgery.
Outcomes and Challenges
Advances in recombinant clotting factor therapies and perioperative management protocols have significantly improved the safety of cardiac surgery in hemophilia patients. Nonetheless, balancing anticoagulation to prevent thrombosis in the CPB circuit against bleeding risks requires precise dosing and monitoring.
Challenges include:
- Maintaining adequate anticoagulation during CPB without excessive bleeding.
- Managing hypothermia-induced coagulation changes and interpreting ACT values accordingly.
- Coordinating multidisciplinary care and ensuring availability of clotting factor concentrates and blood products.
Conclusion
Cardiac surgery involving cardiopulmonary bypass in hemophilia patients is feasible and can be safely performed with intensive clotting factor replacement, weight-based heparin dosing (300–400 IU/kg), continuous ACT monitoring with a target >480 seconds, timely protamine reversal, and vigilant temperature management. A multidisciplinary team approach is essential to optimize outcomes. Continued research is needed to develop standardized protocols tailored to patient-specific factors and surgical complexity.
References
- Palla, R., et al. ‘Management of cardiac surgery in a hemophilia A patient: a case report.’ Haemophilia, 2017.
- Smith, J., et al. ‘Outcomes of cardiac surgery in patients with hemophilia: a retrospective cohort study.’ Annals of Thoracic Surgery, 2019.
- Gandhi, S., et al. ‘Influence of hypothermia on activated clotting time during cardiopulmonary bypass.’ Journal of Cardiothoracic Anesthesia, 2019.
- Kobrinsky, N.L. ‘Hemophilia and cardiac surgery: perioperative management.’ Seminars in Thrombosis and Hemostasis, 2016.
- World Federation of Hemophilia. Guidelines for the Management of Hemophilia. 2020 edition.
- Shann, K., et al. ‘Temperature management during cardiopulmonary bypass: implications on coagulation.’ Annals of Cardiac Surgery, 2018.”
Stay updated with Hemostasis Today.
-
May 12, 2026, 16:46Tagreed Alkaltham: Why Apheresis Matters in Modern Transfusion Medicine
-
May 12, 2026, 16:37Reinhold Kreutz: Cardiovascular Burden in Acute Intermittent Porphyria Needs Greater Awareness
-
May 12, 2026, 16:33Pablo Corral: The Truth About Very Low LDL-Cholesterol
-
May 12, 2026, 16:24Mildred Lundgren: We Must Talk About the Invisible Causes of Stroke
-
May 12, 2026, 16:17Irene Scala: The Sex Disparities In Access to Acute Stroke Treatments In Italy
-
May 12, 2026, 16:04May Nour: UCLA Health Mobile Stroke Unit Becomes The 1st In The World to Perform mCTA In the Field
-
May 12, 2026, 15:57Leonardo Roever: Prognostic Impact of Lipoprotein(a) and CAR in Elderly Acute Ischemic Stroke Patients
-
May 12, 2026, 15:54Bruno Pougault: Prioritizing Laboratory Tests in Resource-Limited Emergency Care
-
May 12, 2026, 15:37Jennifer Holter Chakrabarty: Supporting the Next Generation of Hematology Researchers

