
Athma Dias: Limitations of PT and APTT in Predicting Thrombotic Risk
Athma Dias, Medical Laboratory Scientist at Teaching Hospital Ratnapura, shared a post on LinkedIn:
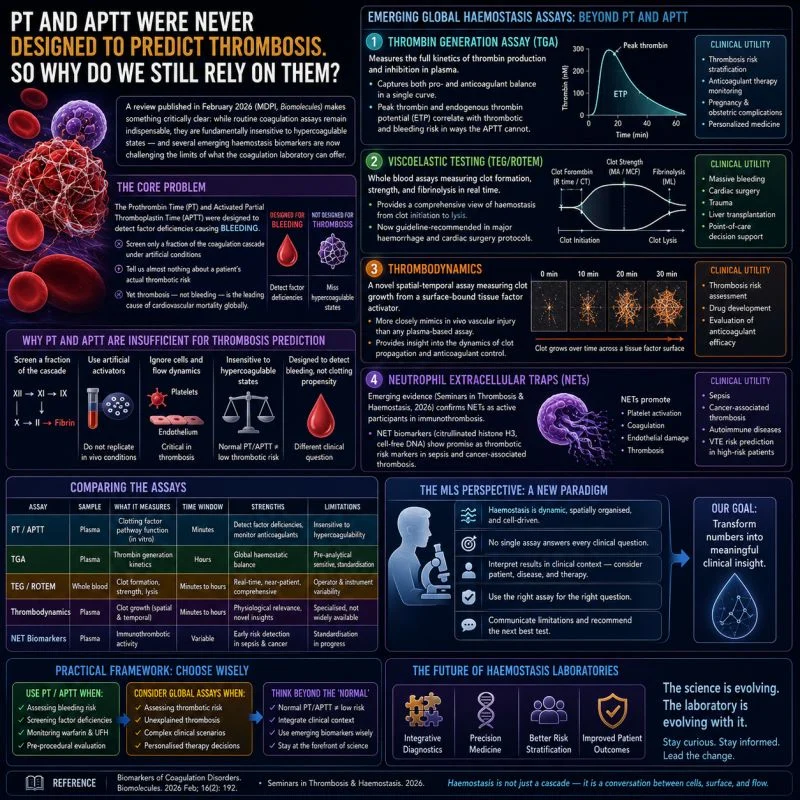
“The PT and APTT were never designed to predict thrombosis.
So why do we still rely on them as if they were?
A review published in February 2026 (MDPI, Biomolecules) makes something critically clear: while routine coagulation assays remain indispensable, they are fundamentally insensitive to hypercoagulable states and several emerging haemostasis biomarkers are now challenging the limits of what the coagulation laboratory can offer.
The core problem:
The Prothrombin Time (PT) and Activated Partial Thromboplastin Time (APTT) were designed to detect factor deficiencies causing Bleeding.
They screen only a fraction of the coagulation cascade under artificial conditions.
They tell us almost nothing about a patient’s actual thrombotic risk.
Yet thrombosis not bleeding is the leading cause of cardiovascular mortality globally.
Emerging global haemostasis assays are addressing this gap:
- Thrombin Generation Assay (TGA) measures the full kinetics of thrombin production and inhibition in plasma, capturing both pro- and anticoagulant balance in a single curve. Peak thrombin and endogenous thrombin potential (ETP) correlate with thrombotic and bleeding risk in ways the APTT cannot.
- Viscoelastic Testing (TEG/ROTEM) whole blood assays measuring clot formation, strength, and fibrinolysis in real time. Now guideline-recommended in major haemorrhage and cardiac surgery protocols.
- Thrombodynamics a novel spatial-temporal assay measuring clot growth from a surface-bound tissue factor activator, more closely mimicking in vivo vascular injury than any plasma-based assay.
- Neutrophil Extracellular Traps (NETs) emerging evidence published in Seminars in Thrombosis and Haemostasis (2026) confirms NETs as active participants in immunothrombosis, with NET biomarkers (citrullinated histone H3, cell-free DNA) showing promise as thrombotic risk markers in sepsis and cancer-associated thrombosis.
For the MLS in haemostasis, this evolution demands more than technical competency on the coagulation analyser.
It demands understanding haemostasis as a dynamic, spatially organised, cell-driven biological process not simply a cascade of clotting factors on a reagent strip.
Reference: Biomarkers of Coagulation Disorders MDPI Biomolecules, February 2026.
Is your laboratory using any global haemostasis assays beyond routine coagulation screening?”
Stay updated with Hemostasis Today.
-
May 8, 2026, 19:37Mostafa Faisal Mohammed Saleh: Exploring the Role of AI and Learning in Monitoring Iron Overload in Beta-Thalassemia
-
May 8, 2026, 19:27Spelile Mutupira: Hemostasis – How the Body Stops Bleeding Naturally
-
May 8, 2026, 19:15Not All TF Activity Assays Measure Tissue Factor – RPTH Journal
-
May 8, 2026, 18:48Carlos Penalillo Pimentel: Detection of Rare Blood Donor Phenotypes During Investigation of Serologic Discrepancies
-
May 8, 2026, 18:23How SCD Affects Daily Life in sub-Saharan Africa? – American Society of Hematology
-
May 8, 2026, 18:03Risk of Thrombosis After Stopping Anticoagulation in APS With aPL Negativization -JTH
-
May 8, 2026, 17:35Gurpreet Kaur Sagoo: A Rare Bone Marrow Failure Syndrome That Challenges Early Diagnosis
-
May 8, 2026, 17:31Edward Lee Carter։ Stroke Prevention Starts in the Emergency Department – but Doesn’t End There
-
May 8, 2026, 17:14Hayder Kadhim: Determinants and Outcomes of OAC Discontinuation in Atrial Fibrillation

