
Bastu Odoka: Storage Lesions – What Really Happens to Stored Blood
Bastu Odoka, Transfusion Scientist at Effia-Nkwanta Regional Hospital, shared a post on LinkedIn:
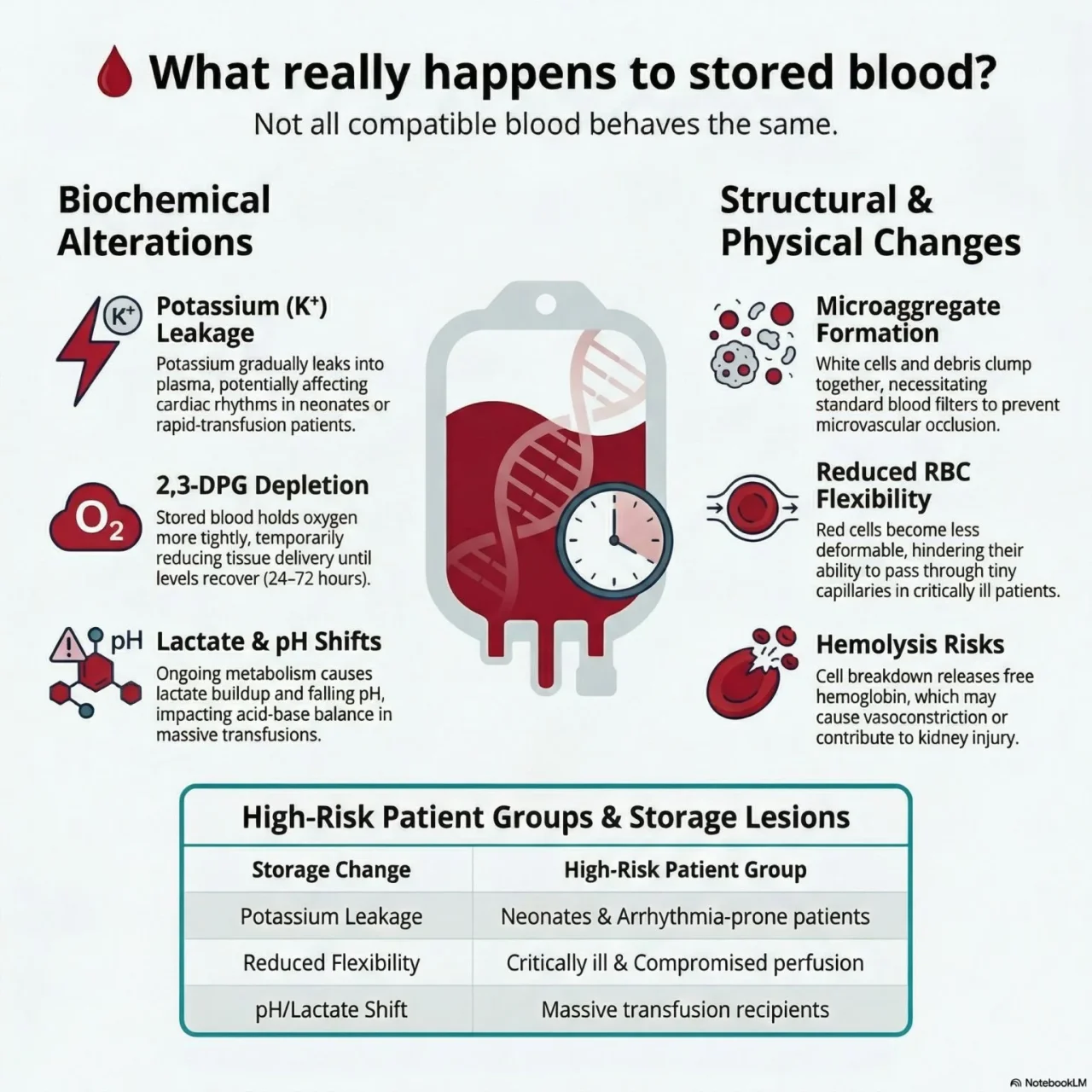
“Storage lesions: what really happens to stored blood
We often focus on compatibility before transfusion.
But there’s another layer we don’t talk about enough, how blood changes during storage.
Over time, stored red cells undergo what we call storage lesions.
Most of the time, this doesn’t change clinical decisions.
But in certain patients, it’s worth paying attention.
1. Potassium leakage
As red cells sit in storage, potassium gradually leaks into the plasma.
This becomes relevant in:
- Neonates
- Patients at risk of arrhythmias
- Rapid or large-volume transfusion
Because high potassium can affect cardiac rhythm.
2. Loss of 2,3-DPG
Stored blood loses 2,3-DPG, which helps haemoglobin release oxygen.
So early after transfusion:
- Haemoglobin holds oxygen more tightly
- Tissue oxygen delivery may be reduced
This matters most in patients who need immediate oxygen delivery.
Good thing: levels recover within 24–72 hours after transfusion.
3. Microaggregate formation
With time, white cells, platelets, and cellular debris aggregate together.
This adds to the particulate load in blood and has been linked to concerns about microvascular flow, may cause pulmonary microvascular occlusion
This is one reason why:
- Leukoreduction
- Standard blood filters
are important in routine practice.
4. Reduced red cell flexibility
Stored red cells become less deformable.
Meaning: They don’t pass through tiny capillaries as easily, which can affect microcirculation in vulnerable patients (critically ill, neonates, and those with already compromised perfusion.)
5. Hemolysis and free haemoglobin
Some red cells break down during storage, releasing free haemoglobin.
This can:
- Bind nitric oxide, causing vasoconstriction
- Contribute to oxidative stress
In certain settings, it may also play a role in kidney injury.
6. Lactate accumulation and pH changes.
Red cells continue to metabolise during storage.
Result:
- Lactate builds up
- pH falls
In most patients, this is well tolerated.
But in large-volume transfusion, especially in:
- Critically ill patients
- Neonates
it can contribute to acid–base imbalance.
So what does this mean in practice?
For the majority of patients, standard-issue blood is safe and appropriate.
These changes are not a reason to avoid transfusion or reject older blood.
But in specific situations, critically ill patients, neonates, massive transfusion, being aware of storage-related changes can help guide safer decisions.
Blood saves lives.
But understanding how it changes helps us use it more thoughtfully.”
Other posts featuring Bastu Odoka on Hemostasis Today.
-
May 24, 2026, 17:48Flora Peyvandi: Crucial Role of Placental ADAMTS13 in Perinatal Survival
-
May 24, 2026, 17:13Mahtab Mashayekhi: The Evolving Role of Red Blood Cell Exchange in the Management of SCD
-
May 24, 2026, 17:12Silvina Graña: Helping Families Better Understand Hemophilia and Daily Life With Bleeding Disorders
-
May 24, 2026, 17:11Miikka Vikkula: Key Insights from the ISSVA 2026 World Congress
-
May 24, 2026, 17:10Mathieu Jackson: Proud to Be Present at This Historic Moment for the Global Bleeding Disorders Community
-
May 24, 2026, 17:09Ramina Fazeli: Highlighting the Emerging Role of Neutrophil Extracellular Traps in Ovarian Cancer
-
May 24, 2026, 16:12William Wallace: Normal B12 Levels May Still Cause Brain Damage and Cognitive Decline
-
May 24, 2026, 15:54Marc Carrier: Ottawa Race Weekend Raised Over 7,000 USD for Thrombosis Canada
-
May 24, 2026, 15:44Janak Sadhu: Building a Stroke-Free Mehsana Through Community Awareness and Healthcare Collaboration

