Fauziya Naikar: Why the MTHFR Gene is a Critical Piece of the Fertility Puzzle
Fauziya Naikar, Junior Embryologist at Star IVF and Fertility Center, shared a post on LinkedIn:
“Why the MTHFR Gene is a Critical Piece of the Fertility Puzzle?
In the embryology lab, we spend our days perfecting morphokinetics and grading.
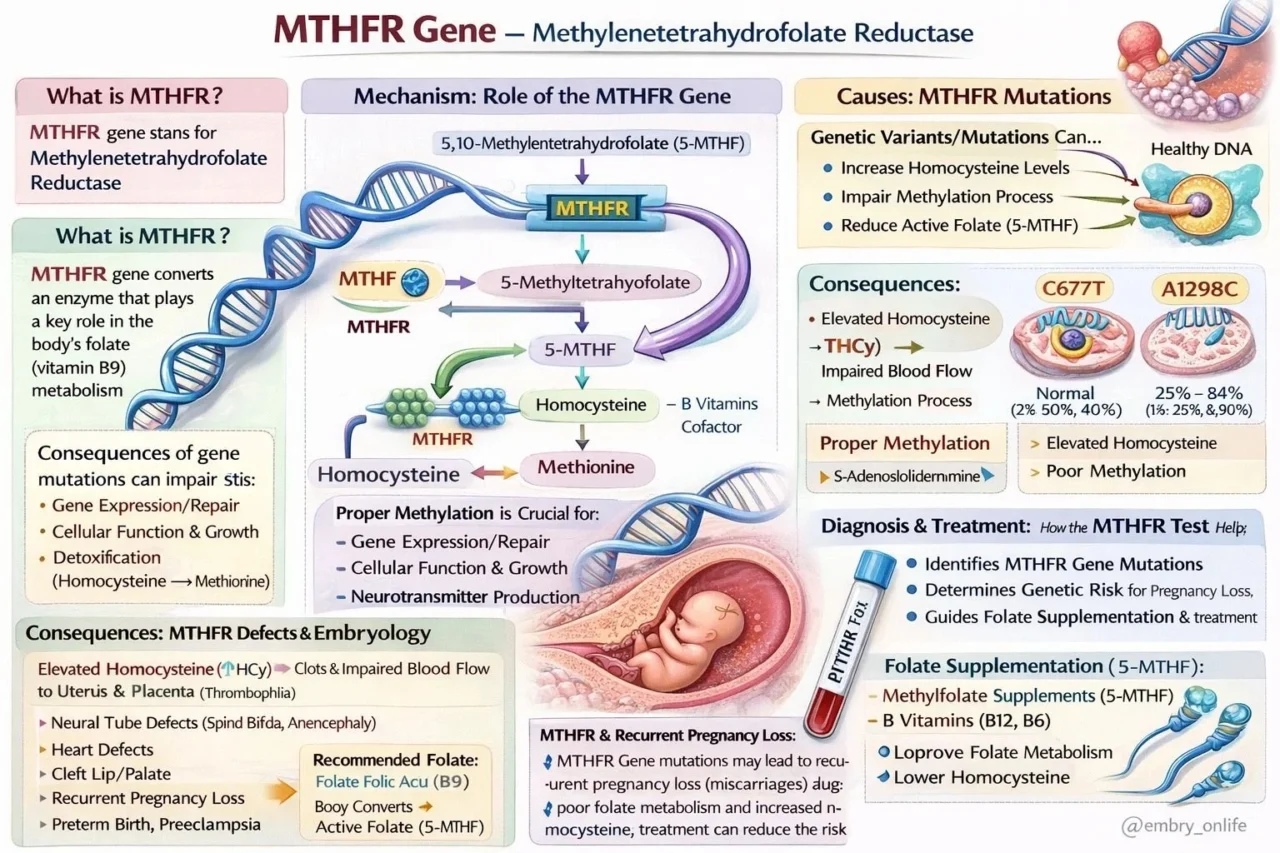
But beyond the petri dish, a ‘silent architect’ is at work: the MTHFR (Methylenetetrahydrofolate Reductase) gene.
Why do high-quality embryos sometimes fail to result in a live birth?
The answer often lies in the biochemical environment provided by parental genetics—specifically the methylation pathway.
The Molecular Breakdown.
The MTHFR enzyme is the critical rate-limiting step in the folate cycle.
Its primary job is to catalyze the conversion of folate into 5-methyltetrahydrofolate (5-MTHF).
This active form is the ‘fuel’ for two vital processes:
- DNA Methylation: This is the cornerstone of epigenetic programming during early cleavage stages. Without it, cellular repair and gene expression are compromised.
- Homocysteine Regulation: MTHFR converts homocysteine into methionine. When mutations like C677T or A1298C occur, enzyme efficiency can drop by 30 percent to 70 percent, causing homocysteine levels to spike.
The Embryologist’s Perspective: Why It Matters?
We see the effort it takes to create a viable embryo, but that embryo needs a stable environment to thrive post-transfer.
Elevated homocysteine (Hyperhomocysteinemia) is a known risk factor for micro-clots in the placental vasculature (Thrombophilia).
This can lead to:
- Implantation failure or early pregnancy loss.
- Neural tube defects (Spina Bifida, Anencephaly).
- Impaired blood flow to the uterus and placenta.
Clinical Pivot: From ‘Unexplained’ to ‘Targeted’.
The traditional reliance on synthetic Folic Acid may not be enough for patients with these genetic variants, as their bodies cannot efficiently convert it.
The Solution?
Bypassing the genetic ‘roadblock’ by using bioavailable Methylfolate (5-MTHF) along with Vitamins B6 and B12.
This targeted approach lowers homocysteine and optimizes the metabolic environment for the embryos we work so hard to cultivate.
By understanding these genomic markers, we move away from the frustration of ‘unexplained infertility’ and toward a more precise, personalized fertility protocol.
Stay updated with Hemostasis Today.
-
May 10, 2026, 16:20Joshua Ngimbwa: How Sex and Socioeconomic Factors Influence Stroke Outcomes in Tanzania
-
May 10, 2026, 16:18Gustavo Monnerat: How Thousands of Fake References Slipped Into Biomedical Literature
-
May 10, 2026, 16:13Dalius Jatužis: Our AI-Driven Automated Stroke Registry Data Extraction Gets Best Poster Award at ESOC 2026
-
May 10, 2026, 16:11Full House for Breakfast with the Experts – American Society for Apheresis
-
May 10, 2026, 15:59Luma Mahairi: AI and Digital Hematology Are Transforming Emergency Laboratory Care in 2026
-
May 10, 2026, 15:55Reza Shojaei: Designing Blood Systems for Chronic Demand
-
May 10, 2026, 15:52Alan Nurden: How Platelets Construct a Thrombus Under Flow
-
May 10, 2026, 15:36Manoj Khadka: Omega-3s and Cardiovascular Outcomes in Hemodialysis
-
May 10, 2026, 15:33Abdul Mannan: Here’s when Your POC INR Machine Misleads You

