
Dhinesh Selvaraju: FDA Approves Narsoplimab (Yartemlea) as 1st Treatment for TA-TMA in Adults and Children
Dhinesh Selvaraju, Clinical Research Assistant at Yuva Intern by Henry Harvin, shared on LinkedIn:
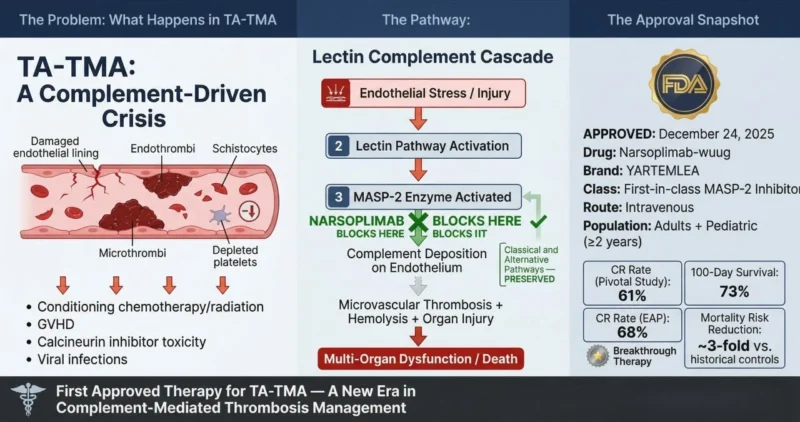
”Hematopoietic stem cell transplant-associated thrombotic microangiopathy (TA-TMA) is a life-threatening complication of allogeneic HSCT characterized by:
- Endothelial injury and microvascular thrombosis
- Microangiopathic hemolytic anemia
- Consumptive thrombocytopenia
- Progressive multi-organ dysfunction
On December 24, 2025, the U.S. Food and Drug Administration approved narsoplimab-wuug (Yartemlea; Omeros Corporation) – the first and only approved treatment for TA-TMA in adults and pediatric patients aged two years and older.
MECHANISM OF ACTION
Narsoplimab-wuug is a first-in-class human IgG4 monoclonal antibody targeting MASP-2 (Mannan-binding lectin-Associated Serine Protease-2).
How it works:
- Selectively inhibits MASP-2, the primary effector enzyme of the lectin complement pathway
- Blocks lectin pathway-mediated complement deposition on damaged microvascular endothelium
- Interrupts the downstream cascade of thrombus formation, platelet consumption, and hemolysis
- Critically preserves the classical and alternative complement pathways, maintaining essential host immune defense in immunocompromised transplant recipients
Triggers of lectin pathway activation in TA-TMA include:
- Transplant conditioning regimens
- Graft-versus-host disease (GVHD)
- Calcineurin inhibitor toxicity
- Viral infections post-transplant
CLINICAL EVIDENCE
Approval was based on a single-arm, open-label pivotal study supplemented by an expanded access program (EAP).
Pivotal Study (n = 28 adult patients):
- TMA complete response (CR) rate: 61%
- 100-day survival rate from TA-TMA diagnosis: 73%
- Comparator analysis: approximately 3-fold lower mortality risk vs external historical control cohorts
Expanded Access Program (n = 19 evaluable patients; 6 pediatric, 13 adult):
- CR rate: 68%
- 100-day survival rate: 74%
SAFETY AND MONITORING
Key adverse reactions:
- Serious infections — most clinically significant risk, consistent with the immunocompromised transplant population
- Monitoring parameters: CBC, LDH, RFT
Notable: The FDA did not mandate a Boxed Warning or REMS for Yartemlea.
Full prescribing information should be reviewed prior to clinical use.
WHY THIS APPROVAL MATTERS
This approval represents a paradigm shift in TA-TMA management.
For decades, clinicians relied solely on:
- Calcineurin inhibitor dose reduction (first-line supportive measure)
- Plasma exchange (limited evidence, no approval)
- Defibrotide (off-label use, no formal TA-TMA indication)
REFERENCES
1. U.S. Food and Drug Administration. FDA approves narsoplimab-wuug (Yartemlea) for hematopoietic stem cell transplant-associated thrombotic microangiopathy. December 24, 2025.
2. Omeros Corporation.”
Stay updated with Hemostasis Today.
-
May 24, 2026, 17:48Flora Peyvandi: Crucial Role of Placental ADAMTS13 in Perinatal Survival
-
May 24, 2026, 17:13Mahtab Mashayekhi: The Evolving Role of Red Blood Cell Exchange in the Management of SCD
-
May 24, 2026, 17:12Silvina Graña: Helping Families Better Understand Hemophilia and Daily Life With Bleeding Disorders
-
May 24, 2026, 17:11Miikka Vikkula: Key Insights from the ISSVA 2026 World Congress
-
May 24, 2026, 17:10Mathieu Jackson: Proud to Be Present at This Historic Moment for the Global Bleeding Disorders Community
-
May 24, 2026, 17:09Ramina Fazeli: Highlighting the Emerging Role of Neutrophil Extracellular Traps in Ovarian Cancer
-
May 24, 2026, 16:12William Wallace: Normal B12 Levels May Still Cause Brain Damage and Cognitive Decline
-
May 24, 2026, 15:54Marc Carrier: Ottawa Race Weekend Raised Over 7,000 USD for Thrombosis Canada
-
May 24, 2026, 15:44Janak Sadhu: Building a Stroke-Free Mehsana Through Community Awareness and Healthcare Collaboration

