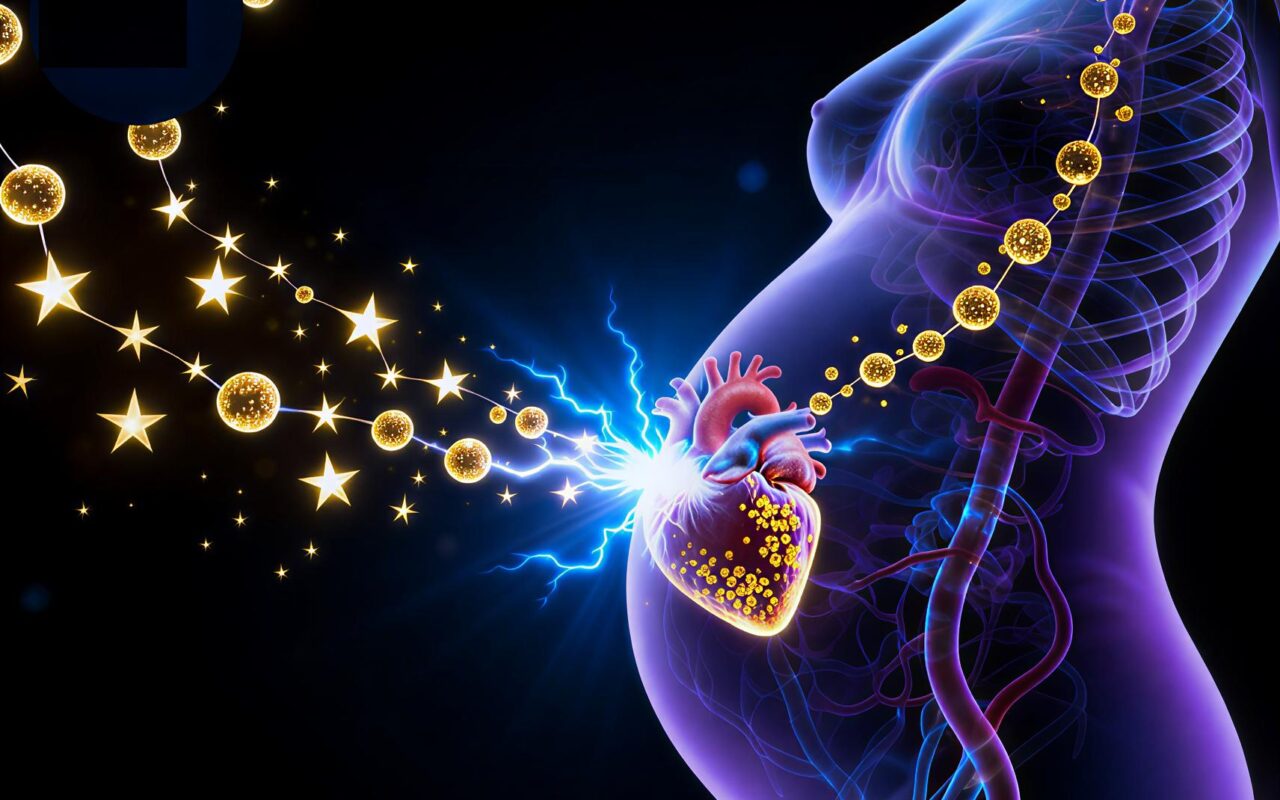
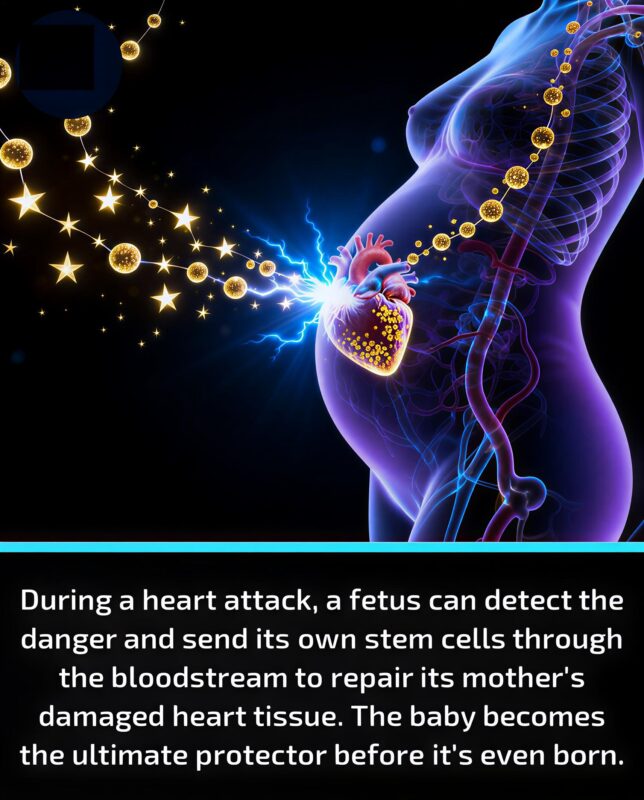
Fetal Stem Cells Repair Mother’s Heart – Xerxes Wennerstierna on the Miraculous Science of Pregnancy
Xerxes Wennerstierna, Book Author AI at A-ZQ, posted on LinkedIn:
”A baby just saved its mother’s life from inside the womb.
That sounds like clickbait. But the biology behind that headline is very real – at least in animal models – and it is changing how we think about pregnancy, regeneration, and long-term female health.
Here’s the short version:
During pregnancy, fetal stem cells can cross the placenta, enter the mother’s bloodstream, and home to injured tissue – including the heart after a heart attack. Once there, they can differentiate into heart muscle, blood vessel, and support cells and actively participate in repair.
In landmark mouse studies from Mount Sinai, researchers induced heart attacks in pregnant females and tracked fetal cells tagged with green fluorescent protein. Those fetal cells:
Crossed into the maternal circulation
Migrated specifically to the damaged area of the heart
Became cardiomyocytes, endothelial cells, and smooth muscle cells in the injury zone
Were associated with better structure and function of the injured hearts compared to controls
This phenomenon is part of fetomaternal microchimerism – the long-term presence of a small population of fetal cells inside the mother’s organs, sometimes decades after pregnancy. These microchimeric cells have been found in maternal heart, brain, lung, and other tissues, and may influence repair, immunity, and even cancer risk.
The story is nuanced: some newer data suggest microchimeric cells can be beneficial in some contexts and potentially neutral or even detrimental in others. Biology is not a Disney script – it is a dynamic, context-dependent ecosystem.
What makes this exciting for medicine is translation:
Scientists have now isolated specific placenta-derived stem cells (Cdx2+ cells) and shown that, when delivered to non-pregnant mice after a heart attack, they can home to the injury site, rebuild tissue, and improve cardiac function. This is the same lineage of cells that appears to traffic naturally from fetus to mother.
We are still at the early stages, but the direction of travel is clear:
Pregnancy is not just “load” on the mother; it may also install long-term cellular protections.
The mother–baby relationship operates on a deep, bidirectional cellular network we’re only beginning to map.
Understanding those signals could unlock new regenerative therapies for heart disease and other organ damage – for men and women, pregnant or not.
Peer-reviewed reading:
Kara RJ et al., Circulation Research – “Fetal cells traffic to injured maternal myocardium and undergo cardiac differentiation”
Mount Sinai summary of the original study
Vadakke-Madathil – “Multipotent fetal-derived Cdx2 cells from placenta” ”
Keep up with Hemostasis Today.
-
May 24, 2026, 17:48Flora Peyvandi: Crucial Role of Placental ADAMTS13 in Perinatal Survival
-
May 24, 2026, 17:13Mahtab Mashayekhi: The Evolving Role of Red Blood Cell Exchange in the Management of SCD
-
May 24, 2026, 17:12Silvina Graña: Helping Families Better Understand Hemophilia and Daily Life With Bleeding Disorders
-
May 24, 2026, 17:11Miikka Vikkula: Key Insights from the ISSVA 2026 World Congress
-
May 24, 2026, 17:10Mathieu Jackson: Proud to Be Present at This Historic Moment for the Global Bleeding Disorders Community
-
May 24, 2026, 17:09Ramina Fazeli: Highlighting the Emerging Role of Neutrophil Extracellular Traps in Ovarian Cancer
-
May 24, 2026, 16:12William Wallace: Normal B12 Levels May Still Cause Brain Damage and Cognitive Decline
-
May 24, 2026, 15:54Marc Carrier: Ottawa Race Weekend Raised Over 7,000 USD for Thrombosis Canada
-
May 24, 2026, 15:44Janak Sadhu: Building a Stroke-Free Mehsana Through Community Awareness and Healthcare Collaboration

