
Dhinesh Selvaraju: A Step Closer to the ‘Ideal Anticoagulant’?
Dhinesh Selvaraju, Clinical Research Assistant at Yuva Intern by Henry Harvin, shared a post on LinkedIn:
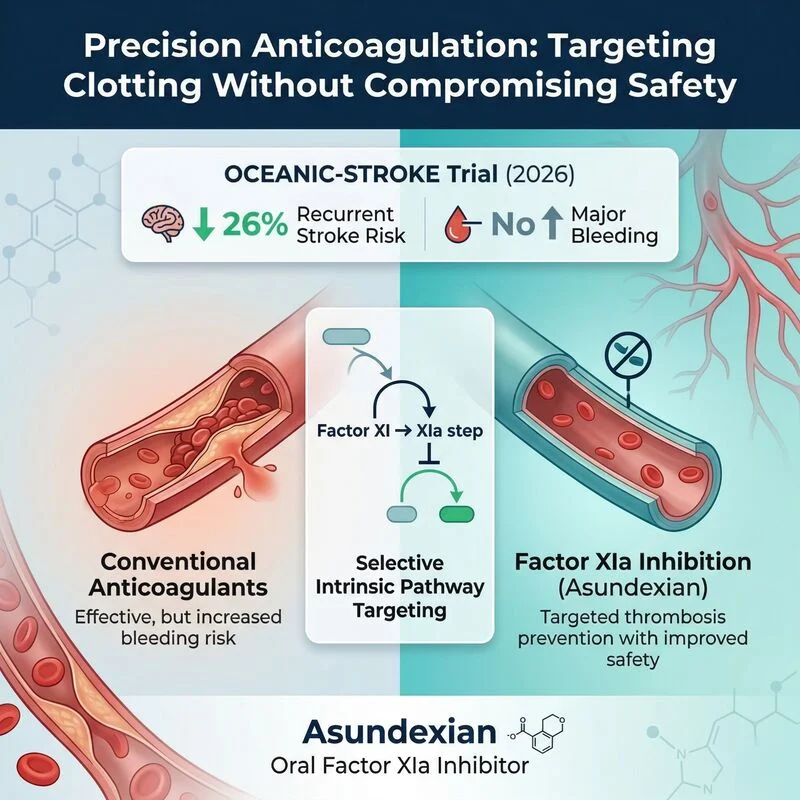
“A Step Closer to the ‘Ideal Anticoagulant’?
A significant advancement in thrombosis research is emerging from recent clinical evidence.
Bayer has reported encouraging Phase III results for its investigational oral Factor XIa inhibitor, Asundexian, evaluated in the OCEANIC-STROKE trial (2026) involving over 12,000 patients.
Key findings:
- Approximately 26% reduction in recurrent stroke risk
- No significant increase in major bleeding events
Why this matters ?
Despite their proven efficacy, current anticoagulants (DOACs, warfarin) are inherently limited by the risk of bleeding complications, often creating a therapeutic trade-off.
Asundexian introduces a novel mechanism:
- Selective inhibition of Factor XIa (intrinsic coagulation pathway)
- Targeting pathological thrombosis while preserving physiological hemostasis
Clinical significance:
- Reinforces the potential of Factor XI inhibition as a safer anticoagulation strategy
- Represents a promising shift in secondary stroke prevention
- Addresses a long-standing clinical challenge: Can thrombosis be prevented without increasing bleeding risk?
A paradigm shift in hemostasis:
This development reflects a broader transition in anticoagulant therapy:
From broad-spectrum anticoagulation to pathway-specific precision targeting
If validated through ongoing regulatory review, asundexian could redefine clinical practice in cerebrovascular risk management and set a new benchmark for next-generation anticoagulants.”
Stay updated with Hemostasis Today.
-
May 12, 2026, 16:46Tagreed Alkaltham: Why Apheresis Matters in Modern Transfusion Medicine
-
May 12, 2026, 16:37Reinhold Kreutz: Cardiovascular Burden in Acute Intermittent Porphyria Needs Greater Awareness
-
May 12, 2026, 16:33Pablo Corral: The Truth About Very Low LDL-Cholesterol
-
May 12, 2026, 16:24Mildred Lundgren: We Must Talk About the Invisible Causes of Stroke
-
May 12, 2026, 16:17Irene Scala: The Sex Disparities In Access to Acute Stroke Treatments In Italy
-
May 12, 2026, 16:04May Nour: UCLA Health Mobile Stroke Unit Becomes The 1st In The World to Perform mCTA In the Field
-
May 12, 2026, 15:57Leonardo Roever: Prognostic Impact of Lipoprotein(a) and CAR in Elderly Acute Ischemic Stroke Patients
-
May 12, 2026, 15:54Bruno Pougault: Prioritizing Laboratory Tests in Resource-Limited Emergency Care
-
May 12, 2026, 15:37Jennifer Holter Chakrabarty: Supporting the Next Generation of Hematology Researchers

