
Ifeanyichukwu Ifechidere: Breaking the INR Myth in Liver Disease
Ifeanyichukwu Ifechidere, Specialist Biomedical Scientist at Sheffield Teaching Hospitals NHS Foundation Trust, shared a post on LinkedIn:
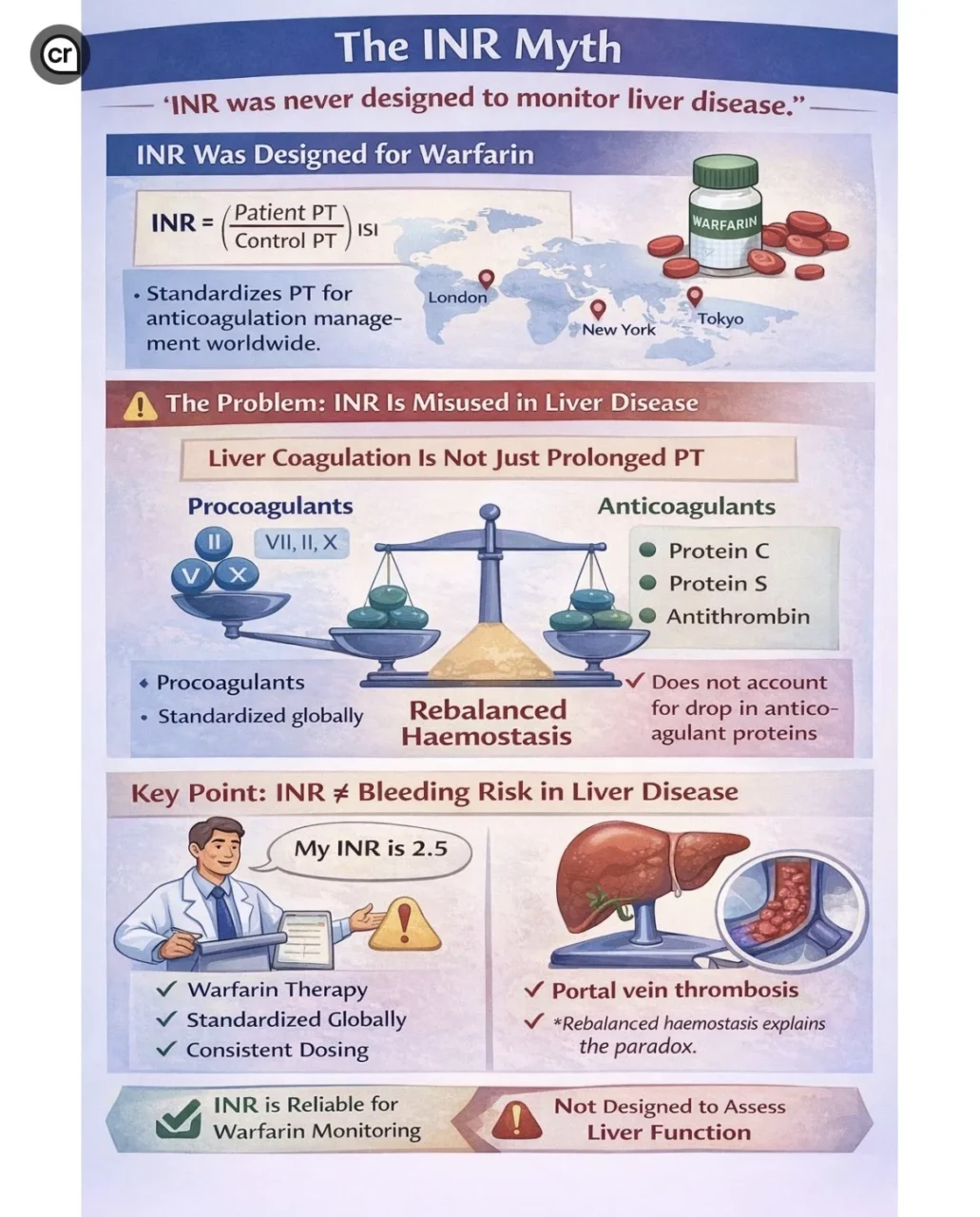
“The INR Myth: ‘INR was never designed to monitor liver disease.’
As biomedical scientists, we see it all the time in lab requests and clinical discussions:
‘The patient’s INR is 2.1 — their liver function must be really poor.’
But here’s the key point many people miss:
INR was never designed to assess liver disease.
It was designed for one specific purpose — standardizing prothrombin time (PT) for patients on warfarin therapy.
Understanding this distinction changes how we interpret coagulation results.
Why INR Exists
Before INR was introduced, Prothrombin Time (PT) results varied significantly between laboratories because different thromboplastin reagents produced different clotting times.
This made warfarin monitoring inconsistent and unsafe.
To solve this, the International Normalized Ratio (INR) was created to standardize PT results across laboratories.
The formula adjusts PT based on the International Sensitivity Index (ISI) of the reagent used.
The goal?
Ensure that a patient on warfarin in London has the same therapeutic interpretation as a patient in New York or Tokyo.
INR was never intended to standardize clotting results in liver disease.
Why INR Can Be Misleading in Liver Disease
In liver disease, the coagulation system becomes far more complex than simply ‘prolonged clotting.’
Yes, the liver produces many procoagulant factors (II, VII, IX, X).
But it also produces anticoagulant proteins such as:
- Protein C
- Protein S
- Antithrombin
In liver failure, both sides of the coagulation balance fall.
This creates what we call ‘rebalanced haemostasis.’
The problem?
INR only reflects reduced procoagulant factors.
It does not measure the parallel drop in anticoagulant proteins.
So a prolonged INR in liver disease does not reliably predict bleeding risk.
What This Means in Practice
A patient with advanced liver disease may have:
An INR of 2.5
Yet still develop portal vein thrombosis
Why?
Because haemostasis in liver disease is dynamic and fragile, not simply ‘auto-anticoagulated.’
This is why relying on INR alone to assess bleeding risk — especially before procedures — can be misleading.
The Bigger Lesson for Laboratory Medicine
Numbers are powerful — but context is everything.
INR is excellent for: Warfarin monitoring
But limited for: Liver disease coagulation assessment
As biomedical scientists, our role is to ensure results are interpreted within the right physiological context.
Because the danger isn’t just abnormal results.
Sometimes it’s misunderstanding what the test was designed to measure.
Do you see INR misused in your setting?
Is it often interpreted as a direct marker of bleeding risk in liver disease?
I’d be interested to hear how different labs and clinicians approach this”
Stay updated with Hemostasis Today.
-
May 12, 2026, 16:46Tagreed Alkaltham: Why Apheresis Matters in Modern Transfusion Medicine
-
May 12, 2026, 16:37Reinhold Kreutz: Cardiovascular Burden in Acute Intermittent Porphyria Needs Greater Awareness
-
May 12, 2026, 16:33Pablo Corral: The Truth About Very Low LDL-Cholesterol
-
May 12, 2026, 16:24Mildred Lundgren: We Must Talk About the Invisible Causes of Stroke
-
May 12, 2026, 16:17Irene Scala: The Sex Disparities In Access to Acute Stroke Treatments In Italy
-
May 12, 2026, 16:04May Nour: UCLA Health Mobile Stroke Unit Becomes The 1st In The World to Perform mCTA In the Field
-
May 12, 2026, 15:57Leonardo Roever: Prognostic Impact of Lipoprotein(a) and CAR in Elderly Acute Ischemic Stroke Patients
-
May 12, 2026, 15:54Bruno Pougault: Prioritizing Laboratory Tests in Resource-Limited Emergency Care
-
May 12, 2026, 15:37Jennifer Holter Chakrabarty: Supporting the Next Generation of Hematology Researchers

