
Niraj Sharma: CHAMPION-AF, CLOSURE-AF, PRAGUE-17, and Why LAAC Still Does Not Dethrone Contemporary Anticoagulation
Niraj Sharma, Cardiac Electrophysiologist/Cardiologist at Northside Hospital, shared on LinkedIn:
“Welcome back – and thank you for being here – for a Breaking News Special Edition of EP Edge Journal Watch.
The late – March LAAC literature now gives us three randomized reference points – CHAMPION – AF, CLOSURE – AF, and PRAGUE – 17 with long – term follow – up – plus a contemporaneous DOAC – safety signal from COBRRA that matters when we interpret what ‘medical therapy’ really means.
Taken together, these data do not justify a broad migration from contemporary anticoagulation toward routine first – line percutaneous left atrial appendage closure in DOAC – eligible atrial fibrillation.
The more defensible reading is narrower:
- LAAC remains a selective tool for specific patients with anticoagulation intolerance, recurrent bleeding, or other compelling constraints – not a general replacement strategy.
At – a – Glance Trial Comparison
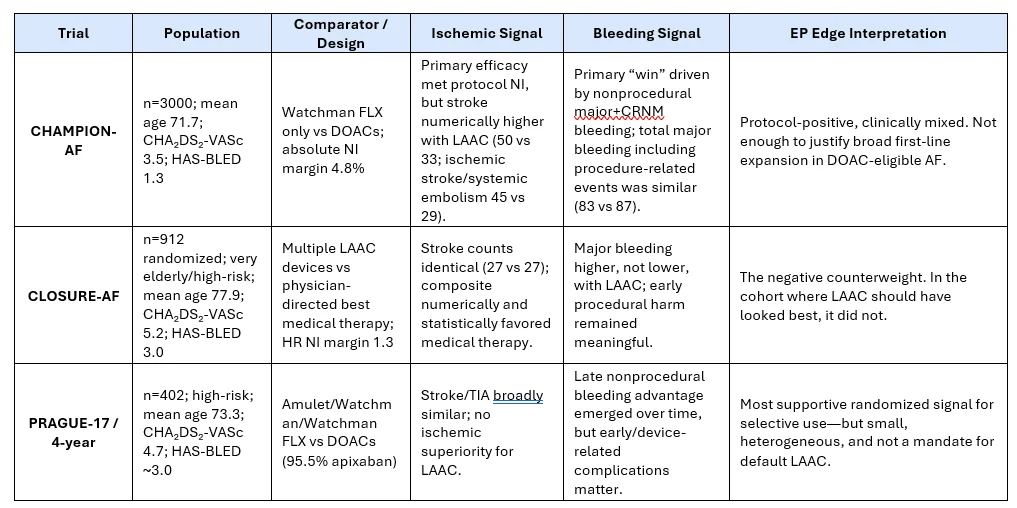
Why this matters:
The three randomized trials are not interchangeable.
They used materially different populations, devices, endpoints, bleeding definitions, and comparator strategies.
Any simple headline that ‘LAAC matches anticoagulation’ misses the design asymmetry.
1. Left atrial appendage closure or anticoagulation for atrial fibrillation (CHAMPION – AF)
Clinical Context
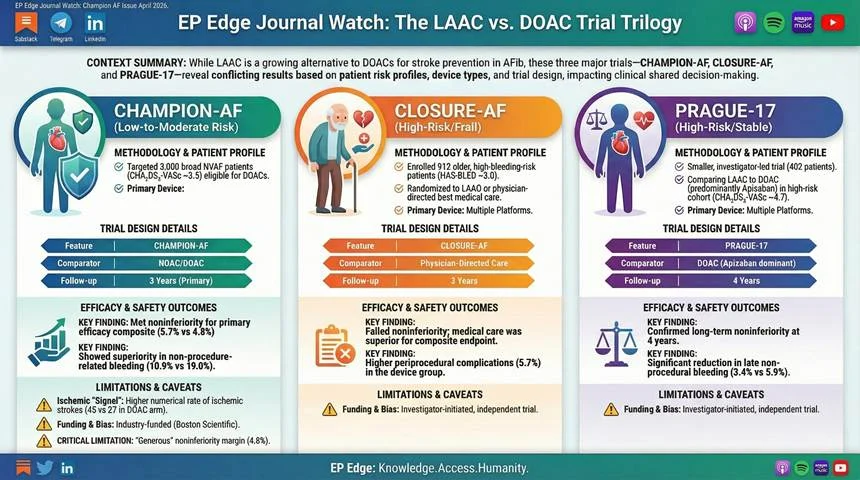
CHAMPION – AF was designed to challenge the default assumption that percutaneous LAAC should remain limited to patients with a rationale to avoid long – term anticoagulation. Unlike CLOSURE – AF, it tested a lower – risk, more procedure – eligible population and used a single contemporary device platform – Watchman FLX.
Study Rationale
The central question was whether Watchman FLX could function not merely as a bailout strategy, but as a first – line alternative to NOAC therapy in patients who were suitable candidates for anticoagulation.
Methodology
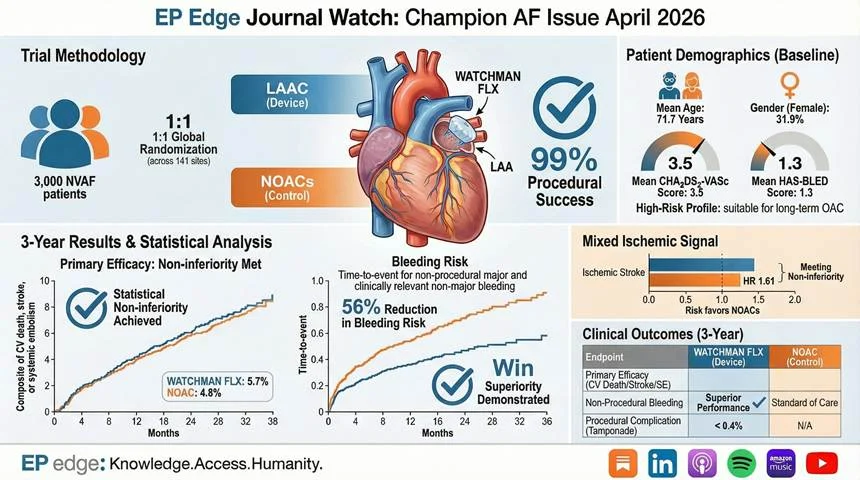
- Prospective, international randomized trial across 141 sites in 16 countries; 1499 patients were assigned to device therapy and 1501 to NOAC therapy.
- Mean age 71.7 years; 31.9% women; mean CHA₂DS₂ – VASc score 3.5; mean HAS – BLED score 1.3. Most patients had paroxysmal AF (68.9%), and 47.8% had undergone prior AF ablation.
- Primary efficacy end point: cardiovascular death, stroke, or systemic embolism at 3 years, tested for noninferiority with an absolute margin of 4.8 percentage points.
- Primary safety end point: non – procedure – related bleeding, defined as ISTH major bleeding plus clinically relevant nonmajor bleeding.
- The device arm was not drug – free early after implantation: 85.0% were discharged on NOAC therapy and 12.6% on dual – antiplatelet therapy.
Key Results
- Primary efficacy end point: 5.7% with LAAC vs 4.8% with NOACs (hazard ratio 1.20; 95% CI 0.87–1.66). The trial met its prespecified noninferiority criterion.
- Overall stroke numerically favored NOAC therapy: 50 events vs 33 events (hazard ratio 1.46; 95% CI 0.94–2.27).
- Ischemic stroke or systemic embolism also numerically favored NOAC therapy: 45 events vs 29 events over 3 years.
- Primary safety end point favored LAAC: non–procedure – related bleeding occurred in 10.9% vs 19.0% (hazard ratio 0.55; 95% CI 0.45–0.67).
- But when major bleeding was counted in a more comprehensive way – procedure – related plus non–procedure – related ISTH major bleeding – the groups looked similar: 5.9% vs 6.4% (hazard ratio 0.92; 95% CI 0.68–1.24).
- Pericardial effusions requiring intervention occurred in 0.7%. Device – related thrombus was seen in 4.8% of imaged patients; 1.8% had clinically relevant thrombus, and 2 patients had a DRT – related stroke.
Methodologic Stress Test
- This was not the classic extreme – bleeding – risk LAAC clinic population: the mean HAS – BLED score was only 1.3.
- The noninferiority margin was absolute rather than relative. Because the observed control event rate was only 4.8%, the upper confidence – limit device rate implied by the published data is about 7.4%, or roughly 1.54× the control rate – substantially more permissive than the trial’s original 1.4 relative – risk framing based on a 12% expected event rate.
- The primary safety endpoint excluded procedure – related bleeding and included clinically relevant nonmajor bleeding. That framing is not illegitimate, but it is highly favorable to a device strategy that inherently trades a one – time invasive hazard for fewer chronic nuisance/CRNM bleeding events.
- The main publication and available source documents do not provide bleeding or ischemic outcomes broken down by specific DOAC agent. That prevents responsible drug – level interpretation against apixaban, rivaroxaban, dabigatran, or edoxaban individually.
Critical Appraisal (EP Edge Take)
Limitations of CHAMPION – AF that materially affect interpretation:
CHAMPION – AF was a large randomized trial, but several design features limit how far its conclusions can be extended.
First, the trial was powered assuming a 3 – year primary – event rate of 12% in each arm and used an absolute noninferiority margin of 4.8 percentage points; the observed event rates were far lower, at 5.7% with device therapy and 4.8% with NOACs, which means the study achieved noninferiority in a setting where the realized event burden was less than half of what had been projected.
In practical terms, when event rates fall that much, an absolute margin becomes more permissive on a relative scale, making it easier to declare ‘noninferiority’ despite numerically worse ischemic outcomes in the device arm.
Second, the primary safety endpoint was constructed as non – procedure – related bleeding, a choice that favors a device strategy by excluding the very upfront hazard intrinsic to implantation.
Once procedure – related major bleeding is included, the apparent bleeding advantage largely contracts, with major bleeding 5.9% in the device arm versus 6.4% in the NOAC arm.
Third, the paper does not provide a meaningful breakdown of the specific NOACs used in the control group, which is a major omission because bleeding risk is not uniform across agents and apixaban in particular has a more favorable safety profile; without that granularity, the magnitude of the device’s ‘bleeding advantage’ is difficult to judge.
Fourth, the trial was sponsor – funded, and the manufacturer collected the data and conducted the analyses, which does not invalidate the findings but does elevate the importance of independent replication.
Fifth, external validity is limited: this was a relatively lower – risk population with mean CHA₂DS₂ – VASc 3.5 and mean HAS – BLED 1.3, so the results do not necessarily generalize to frailer, older, higher – bleeding – risk patients such as those enrolled in CLOSURE – AF.
Finally, important unresolved device – specific issues remain, including device – related thrombus, peri – device leak, and the broader physiologic implications of excluding the left atrial appendage; these are especially relevant because ischemic stroke/systemic embolism numerically favored NOAC therapy and because 5 – year follow – up is still pending.

2. Left atrial appendage closure or medical therapy in atrial fibrillation (CLOSURE – AF)
Clinical Context
If CHAMPION asked whether LAAC could move earlier into lower – risk, DOAC – eligible practice, CLOSURE – AF asked the opposite – and arguably more clinically relevant – question: what happens in older atrial fibrillation patients who are simultaneously at high risk of stroke and high risk of bleeding, the very population in whom LAAC is most often pitched as a solution?
Methodology
- Prospective, multicenter, randomized PROBE trial conducted in Germany; 912 patients were randomized, and the primary analysis included 446 assigned to LAAC and 442 assigned to physician – directed best medical care.
- Mean age 77.9 years; 38.6% women; mean CHA₂DS₂ – VASc score 5.2; mean HAS – BLED score 3.0, reflecting a distinctly older and more fragile cohort than CHAMPION – AF.
- Medical therapy was individualized and included direct oral anticoagulants when eligible, vitamin K antagonists when indicated, and no antithrombotic therapy when contraindicated.
- Primary end point: time to first stroke, systemic embolism, major bleeding, or cardiovascular/unexplained death; noninferiority margin hazard ratio 1.3.
Key Results
LAAC failed noninferiority. A first primary event occurred in 155 LAAC patients versus 127 medical – therapy patients; incidence rates were 16.8 vs 13.3 per 100 patient – years.
- The primary result numerically and statistically leaned toward medical therapy (adjusted hazard ratio 1.28; 95% CI 1.01–1.62).
- Stroke counts were identical in absolute terms: 27 in each arm.
- Major bleeding was not reduced with LAAC: 7.4 vs 6.2 events per 100 patient – years.
- Cardiovascular or unexplained death was numerically higher with LAAC: 9.5 vs 7.7 events per 100 patient – years.
- Periprocedural complications in the device arm remained clinically meaningful, including 18 major bleeding events, 5 tamponades, 2 periprocedural deaths, and 1 device embolization requiring surgery.
Critical Appraisal (EP Edge Take)
CLOSURE – AF is the essential reality check because this is the cohort where LAAC should have looked strongest if the fundamental bleeding – avoidance thesis were broadly correct.
It did not.
Stroke prevention was not better, bleeding was not lower, and the composite end point favored medical therapy.
The implication is uncomfortable but clear: in elderly, high – risk AF patients, procedural vulnerability and early post – LAAC hazard can erase – or even reverse – the theoretical long – term upside.
That result should materially raise the evidentiary bar for interpreting any more favorable LAAC trial.
3. Left atrial appendage closure versus DOACs in high – risk AF (PRAGUE – 17 and 4 – year follow – up)
Why PRAGUE – 17 Still Matters
PRAGUE – 17 remains the most important randomized counterbalance to an overly negative reading of LAAC because it enrolled a genuinely high – risk AF cohort and compared LAAC against a DOAC arm that was overwhelmingly apixaban – based.
Methodology
- Investigator – initiated multicenter randomized noninferiority trial; 402 high – risk AF patients were randomized 1:1 to LAAC or DOAC therapy.
- Mean age 73.3 years; mean CHA₂DS₂ – VASc score 4.7; mean HAS – BLED score about 3.0.
- Entry required either prior bleeding requiring intervention/hospitalization, prior cardioembolism while on anticoagulation, or a combined moderate – high stroke and bleeding profile.
- The DOAC arm was 95.5% apixaban (192 of 201 patients), with dabigatran used in 8 patients and rivaroxaban in 1 patient.
- The LAAC arm was device – heterogeneous: 61.3% Amulet, 35.9% Watchman, and 2.8% Watchman FLX.
- The primary end point was deliberately broad and balanced: stroke/TIA, systemic embolism, cardiovascular death, clinically significant bleeding, or significant procedure – /device – related complications.
Key Results
- Initial results: primary end – point annual rates were 10.99% with LAAC and 13.42% with DOACs; LAAC met noninferiority.
- All stroke/TIA was essentially identical: 2.60% per year with LAAC vs 2.57% per year with DOACs.
- Clinically significant bleeding was numerically lower with LAAC but not statistically different in the initial report (5.50% vs 7.42%; sHR 0.81).
- When device/procedure – related bleeding was excluded, the nonprocedural bleeding signal became more favorable for LAAC (3.76% vs 7.42%; sHR 0.53), although this did not reach significance in the original report.
- At 4 years, the primary noninferiority result persisted, and nonprocedural clinically relevant bleeding became significantly lower with LAAC (3.4% vs 5.9%; sHR 0.55; P=0.039).
- Major LAAC – related complications occurred in 9 patients (4.5%), emphasizing again that the early procedural cost is real.
Critical Appraisal (EP Edge Take)
PRAGUE – 17 is the most supportive randomized evidence for LAAC as a selective alternative in high – risk AF patients, but it should not be oversold.
Its most persuasive long – term advantage is late nonprocedural bleeding reduction, not superior ischemic protection; and its comparator was not ‘DOAC class in the abstract’ but effectively an apixaban – dominant strategy.
That makes PRAGUE clinically important – but not a mandate for default LAAC. It supports careful selection, not automatic substitution.
4. Cross – Trial Synthesis: Where COBRRA 4. Cross – Trial Synthesis: Where COBRRA Fits – and Where It Does NotFits – and Where It Does Not
COBRRA was not an atrial fibrillation trial.
It randomized patients with acute venous thromboembolism to apixaban or rivaroxaban and therefore cannot be used to recalculate AF – specific trade – offs or to prove that one AF comparator arm was ‘too favorable.’
But it does contribute one important interpretive lesson: DOACs are not a monolith.7
- Clinically relevant bleeding at 3 months was 3.3% with apixaban and 7.1% with rivaroxaban (relative risk 0.46; 95% CI 0.33–0.65).
- Major bleeding was 0.4% with apixaban and 2.4% with rivaroxaban.
- Recurrent symptomatic venous thromboembolism was similar (1.1% vs 1.0%).
- The accompanying editorial’s conclusion was blunt: the choice between these two agents for VTE is no longer a toss – up.
Why does that matter here?
Because a device strategy claiming a ‘bleeding advantage over NOACs’ should be judged against the actual NOAC mix.
PRAGUE – 17 explicitly tells us that 95.5% of its DOAC arm was apixaban.
CHAMPION – AF, in contrast, does not report drug – specific bleeding or ischemic outcomes by NOAC type in the main publication or in the provided source files.
That omission matters.
If the comparator was apixaban – heavy, the true practical bleeding hurdle for LAAC is lower than a generic ‘NOAC’ label implies.
If the comparator leaned toward rivaroxaban, a class – level summary may exaggerate what one would observe in an apixaban – first practice environment.
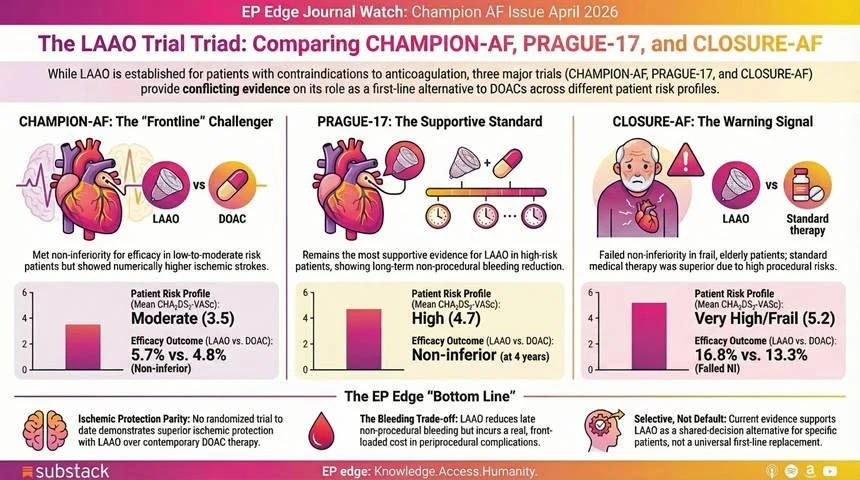
The practical synthesis is this: across CHAMPION – AF, CLOSURE – AF, and PRAGUE – 17, no randomized trial shows superior ischemic protection with LAAC over contemporary anticoagulation.
The bleeding story depends heavily on which events are counted, when they are counted, and which medical comparator is sitting across the table.

5. How This Should Change Practice Today
- Routine first – line LAAC for broadly DOAC – eligible AF patients should not become the new default on the basis of CHAMPION – AF.
- For older, frailer, high – bleeding – risk patients, CLOSURE – AF reinforces that optimized physician – directed medical therapy remains a valid – and often preferable – default strategy.
- LAAC remains reasonable for carefully selected patients with recurrent major bleeding, true anticoagulation intolerance, inability to sustain long – term OAC, or other compelling individualized constraints; PRAGUE – 17 is the strongest randomized support for that narrower position.
- For a patient doing well on apixaban, current evidence does not justify switching to elective percutaneous LAAC simply to avoid pills or to reduce nuisance bleeding.
- The field still needs longer – term data and cleaner comparator reporting – especially drug – specific DOAC breakdowns – before recommendations should be materially expanded.135
EP Edge Bottom Line
- CHAMPION – AF was statistically positive by protocol, but clinically mixed: more ischemic events in the device arm and no convincing reduction in total major bleeding.
- CLOSURE – AF is the harder signal to ignore because it tested the older high – risk patients in whom LAAC is most often justified – and LAAC failed there.
- PRAGUE – 17 supports LAAC as a selective alternative in carefully chosen high – risk patients, especially when long – term nonprocedural bleeding reduction matters, but not as a superior ischemic strategy.
- COBRRA reminds us that DOAC choice matters. ‘NOAC’ is not a single bleeding phenotype, and future LAAC trials must be interpreted against the actual comparator drug mix.
- Net result: DOACs remain the evidence standard in AF. LAAC stays in the toolbox, but as a selective strategy – not a universal escape hatch from anticoagulation.1357
Closing
Thank you for reading EP Edge Journal Watch.
A simultaneous podcast version of this issue is available across podcast platforms, and the full newsletter archive is also available via Substack and LinkedIn.
EP Edge Journal Watch (Weekly Newsletter)
EP Edge Newsletter (In – Depth Monthly Issues)
EP Edge Podcast:
EP Edge Journal Watch Podcast:
Read, Subscribe, Listen.”
Title: Left Atrial Appendage Closure or Anticoagulation for Atrial Fibrillation
Authors: Shephal K. Doshi, Saibal Kar, Devi G. Nair, Thomas Waggoner, Himanshu Agarwal, Mehran Moussavian, Amir Kashani, Saumil Oza, Leon Feldman, Ashish Sadhu, David DeLurgio, Oluseun Alli, Jens Erik Nielsen-Kudsk, Masanori Yamamoto, Mohamad Alkhouli, A. John Camm, Megan Coylewright, C. Michael Gibson, Christopher B. Granger, Mahmut Edip Gurol, Kenneth C. Huber, Moussa Mansour, Andrea Natale, Stuart J. Pocock, Vivek Y. Reddy, Walid I. Saliba, Federico M. Asch, Scott Wehrenberg, Kate Frost, Thomas Christen, Brad S. Sutton, Kenneth M. Stein, Martin B. Leon, Kenneth A. Ellenbogen
Read the Full Article on NEJM

Title: Left Atrial Appendage Closure or Medical Therapy in Atrial Fibrillation
Authors: Ulf Landmesser, Carsten Skurk, Paulus Kirchhof, Thorsten Lewalter, Johannes Hartung, Andi Rroku, Burkert Pieske, Johannes Brachmann, Ibrahim Akin, Claudius Jacobshagen, Benjamin Meder, Andreas Zeiher, Stefan D. Anker, Holger Thiele, Stefan Blankenberg, Steffen Massberg, Heribert Schunkert, Norbert Frey, Alexander Joost, Martin Bergmann, Ralph Stephan von Bardeleben, Tim Friede, Marius Placzek, Anna Suling, Karl Georg Haeusler, Matthias Endres, Karl Wegscheider, Leif-Hendrik Boldt, Ingo Eitel
Read the Full Article on NEJM

Title: Left Atrial Appendage Closure Versus Direct Oral Anticoagulants in High-Risk Patients With Atrial Fibrillation
Authors: Pavel Osmancik, Dalibor Herman, Petr Neuzil, Pavel Hala, Milos Taborsky, Petr Kala, Martin Poloczek, Josef Stasek, Ludek Haman, Marian Branny, Jan Chovancik, Pavel Cervinka, Jiri Holy, Tomas Kovarnik, David Zemanek, Stepan Havranek, Vlastimil Vancura, Jan Opatrny, Petr Peichl, Petr Tousek, Veronika Lekesova, Jiri Jarkovsky, Martina Novackova, Klara Benesova, Petr Widimsky, Vivek Y Reddy
Read the Full Article on JACC

Stay updated on all scientific advances on Hemostasis Today.
-
May 22, 2026, 13:08Anas Hamad: Thrombosis as Overlooked Clinical Challenge in APL
-
May 22, 2026, 12:29Neema Ngugi: Highlighting 6 Major Types of Anemia
-
May 22, 2026, 12:27Jacinto López Sagaseta: Solving the Structural Puzzle of FVIIa–EPCR Interaction
-
May 22, 2026, 12:25Shehab Mohamed: Exploring the Emerging Link Between JAK2 Mutations and Thoracic Aortic Aneurysms
-
May 22, 2026, 11:52Prakash W. Kamat: Highlighting the Role of Physical Activity in Hemophilia Management
-
May 22, 2026, 11:43Mohamed Hanafy Morsy: Understanding Hemochromatosis – Genetics Made Simple
-
May 22, 2026, 10:23Rucha Patil: Insights from The National Haemophilia Conclave 2026
-
May 22, 2026, 10:11Narat Srivali: New Systematic Review and Meta-Analysis on Glucocorticoid Therapy and The Risk of VTE
-
May 22, 2026, 10:02Greta Mulders: Nursing Leadership in Haemophilia Gene Therapy

